
شبیه سازی دینامیک ملکولی غیر تعادلی جریان گاز در کانال های نانو ارگانیک
چکیده
هدف مطالعه حاضر این است که کمک مرحله جذب شده به شار جرم را بررسی کند و انتقال گازها را با کشش های (میل ترکیبی) مختلف جذب سطحی در کانال های آلی مقیاس نانو مقایسه کند. در این مطالعه برای مقایسه انتقال (جابجایی) گازهایی با کشش جذب زیاد (متان و آرگون)، با گازهایی با کشش جذب کم (هلیوم)، از شبیه سازی های دینامیک ملکولی غیر تعادلی نیرو-محور (NEMD) استفاده کرده ایم، که ارتفاع کانال ها 2، 4، 6 و 8 نانومتر (nm) در دو عدد نودسِن به اندازه 0.1 و 0.2 بود. خصوصیات سرعت و شار جرم در بین کانال ها برای آرگون، متان و هلیوم مقایسه می گردد. ضریب های پخش جابجایی و شار ملکولی این گازها نیز محاسبه شده است. خصوصیات جذب (جذب سطحی) هم با استفاده از شبیه سازی های Grand Canonical Monte Carlo تحلیل شده است. برای همه گازهای مطالعه شده، خصوصیات سرعت (به شکل دو شاخه) بدون توجه به اندازه کانال و عدد نودسن، مشاهده شد. خصوصیات شار جرم آرگون و متان در بین کانال ها، کمک مهم ملکول های جذب شده به شار جرم کلی را اثبات می کند. همچنین وقتی که عدد نودسن افزایش می یابد، کمک مرحله جذب شده به شار جرم کلی بیشتر می شود. شار ملکولی هلیوم در همه اندازه های کانال، نسبت به آرگون و متان کوچکتر است. ضریب های پخش محاسبه شده متان در مقایسه با آرگون برای همه اندازه های کانال بزرگتر است، و زمانیکه اندازه کانال افزایش می یابد این ضریب ها کاهش می یابند. وقتی که عدد نودسن افزایش می یابد، ضریب های پخش برای آرگون و متان کوچکتر می گردد. ضریب های پخش برای هلیوم، تابع ضعیفی از اندازه کانال و عدد نودسن است. بر اساس نتایج، کمک ملکول های جذب شده می تواند بیش از 50 درصد شار جرم کلی کانال باشد. برای مطالعات دامنه های فشار، ضریب پخش هلیوم در مقایسه با آرگون و متان، حساسیت کمتری نسبت به فشار و عدد نودسن دارد. این مطالعه نشان می دهد که انتقال از طریق مجراهای آلی مقیاس نانو، ضرورتاً بصورت پخشی (انتشاری) است. بنابراین برای داشتن یک مدل واقع گرایانه برای پیش بینی بازیابی سیال ها (شاره ها) از منابع غیرمتعارف ، باید معادلات انتقال در منفذهای نانوی (ریز منفذها) آلی را با معادلات انتقال پخشی جایگزین کنیم.
1- مقدمه
مکانیزم های بازیابی گاز از منابع سنگ رس بعلت حضور ریزمنفذهای آلی، با مکانیزم های بازیابی گاز از منابع متعارف تفاوت دارند. از یک طرف، ملکول های گاز به جذب شدن به سطوح منفذهای آلی تمایل دارند که این منجر به پخش ناهمگن سیال در منفذها می گردد. جذب ملکول های گاز به سطوح منفذ سبب تأثیرات انتقال سطح ملکولی (مانند پخش سطحی) می شود. از طرف دیگر، اندازه منفذها مشابه مسافت آزاد میانگین ملکول ها است که سبب سیستم های جریان غیر ممتد می گردد. انتقال سیال ها در این سیستم ها را نمی توان توسط معادلات هیدرودینامیک پیوستار-محور قدیمی توصیف کرد. سپس این سوال پیش می آید که انتقال گازهایی با کشش جذب کم (مانند هلیوم) تا چه حد با انتقال گازهایی با کشش جذب زیاد (مانند متان و آرگون) در مجراهای آلی مقیاس نانو تفاوت دارد؟ پاسخ این سوال می تواند درک ما از مکانیزم های انتقال سیال در منابع سنگ رس را افزایش دهد.
4- نتیجه گیری ها
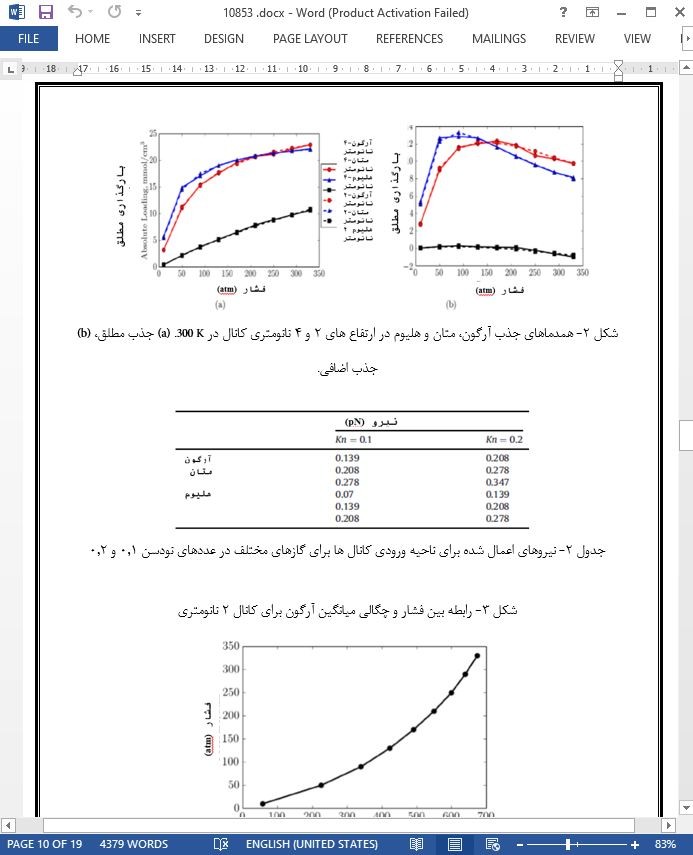
جذب و انتقال نیرو-محور هلیوم، آرگون و متان در چهار کانال گرافیت با ارتفاع های 2، 4، 6 و 8 نانومتری توسط اجرای شبیه سازی های GCMC و NEMD مورد مطالعه قرار گرفته است. متان و آرگون رفتار جذب مشابهی (جذب های مطلق و اضافی) را نشان می دهند درحالیکه چگالی هلیوم در سراسر ارتفاع کانال تقریباً ثابت است. برای همه گازها [چه کشش جذب آنها زیاد باشد (آرگون و متان) و چه کم باشد (هلیوم)]، خصوصیات سرعت به شکل دو شاخه، برای همه ارتفاعات کانال ایجاد شده است. بنابراین بخاطر مقادیر چگالی جذب شده بیشتر آرگون و متان، مرحله جذب شده کمک زیادی به شار جرم کلی در سراسر کانال می کند. خصوصیات شار جرم آرگون و متان در سراسر کانال ها، کمک مهم ملکول های جذب شده به شار جرم کلی را اثبات می کند. طبق این نتایج، وقتی که عدد نودسن افزایش می یابد، کمک انتقال مرحله جذب شده به شار جرم کلی کانال ها بیشتر می شود. این کمک در عدد نودسن 0.1 برای آرگون و متان 42 درصد است، و در عدد نودسن 0.2 به حدود 53 درصد افزایش می یابد.
Abstract
The objective for this work is to investigate the contribution of the adsorbed phase to the mass flux and comparing transport of gases with different adsorption affinities in organic nano-scale channels. In this work, force-driven Non-Equilibrium molecular dynamics (NEMD) simulations are used to compare the transport of gases with high adsorption affinity (Methane and Argon) with the ones with low adsorption affinity (Helium), for channel heights of 2, 4, 6, and 8 nm at two Knudsen numbers of 0.1 and 0.2. Velocity and mass flux profiles across the channel for Argon, Methane, and Helium are compared. Transport diffusion coefficients and molecular flux of these gas are also calculated. Furthermore, adsorption properties are analysed using Grand Canonical Monte Carlo simulations. For all the gases studied, plug-shaped velocity profiles are observed irrespective of the channel size and Knudsen number. Mass flux profiles of Argon and Methane across the channels demonstrate a significant contribution of adsorbed molecules to the total mass flux. Furthermore, as Knudsen number increases, the contribution of the adsorbed phase to the total mass flux becomes higher. Molecular flux of Helium is smaller than that of Argon and Methane for all channel sizes. The calculated diffusion coefficients of Methane are higher than those for Argon for all the channel sizes and they decrease as the channel size increases. For Argon and Methane, the diffusion coefficients become smaller as Knudsen number increases. For Helium, the diffusion coefficients are weak functions of the channel size and Knudsen number. Based on the results, contribution of the adsorbed molecules can be more than 50% of the total mass flux of the channel. For the pressure ranges studies, transport diffusivity of Helium is less sensitive to pressure and Knudsen number compared to Argon and Methane. This study shows that the transport through organic nano-scale conduits is essentially diffusive. Therefore, to have a realistic model for predicting the recovery of fluids from unconventional resources, the transport equations in organic nanopores should be replaced by the diffusive transport equations.
1. Introduction
Gas recovery mechanisms from shale resources are different from those of conventional resources due to presence of organic nanopores. In one hand, the gas molecules have a tendency to be adsorbed to the surfaces of the organic pores that results in heterogeneous fluid distributions within the pores. Adsorption of gas molecules to the pore surfaces causes molecular level transport effects, such as surface-diffusion. On the other hand, the pore sizes are comparable to the mean free path of the molecules, which causes non-continuum flow regimes. The transport of fluids in this systems cannot be described by classical continuum-based hydrodynamic equations. A question then arises as to how different is the transport of gases with low adsorption affinity, e.g., Helium, compared to the ones with high adsorption affinities, e.g., Methane and Argon, in organic nano-scale conduits? Answer to this question can enhance our understanding of the fluid transport mechanisms in shale resources.
4. Conclusions
Adsorption and force-driven transport of Helium, Argon, and Methane in four graphite channels of 2, 4, 6, and 8 nm hight is studied by performing GCMC and NEMD simulations. Methane and Argon show similar adsorption behavior (absolute and excess adsorptions) while the density of Helium across the channel height is approximately constant. For all gases, whether the adsorption affinity is high (Argon and Methane) or low (Helium), plug-shaped velocity profiles are established for all the channel heights. Therefore, due to the higher adsorbed density values of the Argon and Methane, the adsorbed phase contribute significantly to the overall mass flux across the channel. Mass flux profiles of Argon and Methane across the channels demonstrate a significant contribution of adsorbed molecules to total mass flux. According to these results, as Knudsen number increases, the contribution of the adsorbed phase transport to the total mass flux of the channels becomes higher. This contribution is 42% for Argon and Methane at Knudsen number of 0.1, which increases to approximately 53% at Knudsen number of 0.2.
چکیده
1- مقدمه
2- روشهای دینامیک ملکولی
3- نتایج و بحث
3.1- تعادل
3.2- جریان نیرو-محور
4- نتیجه گیری ها
Abstract
1. Introduction
2. Molecular dynamics methods
3. Results and discussion
3.1. Equilibrium
3.2. Force-driven flow
4. Conclusions
- ترجمه فارسی مقاله با فرمت ورد (word) با قابلیت ویرایش، بدون آرم سایت ای ترجمه
- ترجمه فارسی مقاله با فرمت pdf، بدون آرم سایت ای ترجمه
