
دانلود رایگان مقاله طیف سنجی جذب سطحی اتمی کوره فلزی الکترو حرارتی
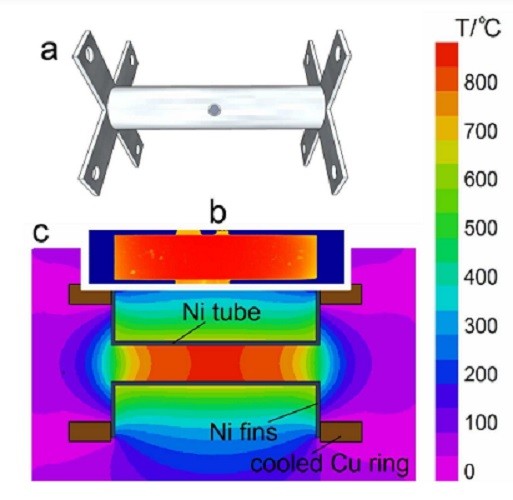
ما سیستم اتم سازی برای طیف سنجی جذب سطحی اتمی در مقایسه با کوره استیل ضد زنگ حرارت داده شده توسط اثر ژول با ابزار مقاومت اصلی آن معرفی می کنیم. نوع جدیدی از کوره نیاز به هر گونه گازی در مدت عملیات ندارد. نمونه با انژکتور اسپری حرارتی بصورت مستقل کنترل شده معرفی می شود. دستگاه با FAAS رسمی عملکرد بهتری (طیف سنجی جذب اتمی شعله) برای آنالیت های زیادی دارد، گزینه ای ارزان، خلاصه و امن را برای دترمینان های تحلیلی زیادی ارائه می دهد. ویژگی کامل سیستم معرفی و شبیه سازی نظری با داده های آزمایشی مقایسه می شود.
مقدمه
طیف سنجی جذب سطحی اتمی (AAS) یکی از گسترده ترین تکنیک ها برای تعین عناصر از زمان معرفی طیف سنجی جذب سطحی اتمی شعله (FAAS) توسط آلن واش در سال 1955 می باشد 1.
از آن زمان، تغیرات زیادی از ایده اصلی بررسی شده است و چندین نسخه جدید متنوعی از AAS با موفقیت پیشنهاد شده است. 2،3
کاربرد شعله در FAAS بر پذیرش دو نواقص اشاره می کند: مجموعه سیستم مدیریتی گاز قادر است تا از عملیات سالم تحت هر شرایطی و زمان کوتاه سکونت نمونه در مسیر نوری 4 مطمئن شود، حقیقت این است که شعله محیط بازی است.
اتم ساز الکتروحرارتی8-5، اکثرا کوره های گرافیت 9،12 یا کویل تنگستن 13-16 در میزان کمتر، گزینه عالی برای غلبه بر مسائل توصیف شده در بالا می باشد. به هرحال، برای گزینه های مذکور و صرفنظر از کاربرد کوره های پلت فرم دمای استاندارد، اصلاحات ماتریکس یا گرمایشی اریب می شود 17،18، جریان گاز آراگون با خالصی بالا برای حفاظت سلول حرارتی اجباری است.
استراتژی دیگری برای محدود کردن نمونه در مسیر نهایی اخیرا توسط برندت19 پیشنهاد و توسط چندین گروه اصلاح شد20-25. این نظم و ترتیب به عنوان طیف سنجی جذب سطحی اتمی کوره شعله اسپری حرارت شناخته شده است (TS-FFAAS)، که به معرفی کلیت نمونه مایع در لوله (یای کوره) نصب شده در احتراق شعله در مسیر نوری طیف سنجی جذب سطحی اتمی اجازه می دهد. نمونه از طریق پمپ کرمکی تزریق می شود و به کوره شعله (FF) انتقال داده شده از طریق موئین سرامیکی مستقیما توسط شعله گرم می شود، که محصول تولید شده "اسپری حرارتی" نامیده می شود(TS). ترکیب معرفی نمونه کل و محدودیت نمونه اتمی برای بهینه سازی حساسیت برای عناصر فرار ثابت شده است زمانیکه FAAS بدون نیاز تغیرات اصلی در ابزار اصلی مقایسه می شود.
در FFAAS و ETAAS تحریک شده، ما رویکرد طیف سنجی جذب سطحی اتمی کوره فلزی الکترو حرارتی (EMFAAS) را معرفی می کنیم که مزیت هر دو تکنیک را دارد. EMFAAS از لوله آلیاژ استیل ضد زنگ به عنوان کوره استفاده می کند. این لوله نسبتا مقاومت الکتریکی بیشتری دارد که بسیار متناسب با حرارت با انجام جریان بالا از طریق بدن آن می باشد.
بنابراین، دمای بالا کوره بدون نیاز به سوخت یا حفاظت از گاز حاصل می شود. معرفی نمونه مایع کل با کمک پمپ کرمکی و انژکتور اسپری حرارتی سرامیک انجام می شود که از نظر الکتریکی گرم و مستقل از حرارت کوره می باشد. در این شیوه، EMFAAS گرمای الکتریکی هر دو انژکتور FF و TS را ترکیب می کند، دور از فشار گاز یا از طریق احتراق، حفاظت یا نمونه های مایع گردشدگی بادی معمولی می باشد.
علاوه براین، EMFAAS ، سادگی و استحکام FAAS را با هم با افزایش حساسیت برای عناصر حیاتی ارائه می دهد همانطورکه آن در اینجا نشان داده خواهد شد.
تجربی
شیوه و مواد
کلیه راه حل ها با واکنشگرهای شیمیایی رتبه تحلیلی و آب دیونیزه دوبل (DDW) حاصل از سیستم خالص سازی Milli-Q آماده شد (میلیپور، بدفورد، کارشناسی ارشد، آمریکا). کلیه ظروف شیشه ای با EXTRAN (Merck) 1% v/v شسته و در 10% (v/v) HCl با تمیزی بیشتری با DDW حفظ شد. راه حل های استاندارد کلیه آنالیت ها با استفاده از رقیق سازی مناسب محلول های ذخیره 1,000 g L-1 آماده شد (مرک دارمس تادت، آلمان). دوربین دیجیتال سامسونگ مدل NX 1100 برای عکس گرفتن از کلیه تصاویر دما استفاده شد. حرارت دادن به سول با ابزار تجاری مبدل اجاق فر مایکروویو 700 W انجام شد که نوشته های فرعی اصلی توسط دو نوبت کویل 7 میلی متر قطر مس کابل های چند فیبره جایگزین شد ( خودرو، برای باطری ها )
طیف سنجی جذب سطحی اتمی شیمادزو AA6800(شیمادزو، کیوتو، ژاپن)، لامپهای توخالی کاتد (هاماماتسو، ژاپن) و لامپ دوتریوم برای اصلاح سابقه در سراسر ارزیابی ها استفاده شد. سایر شرایط ابزاری توسط تولید کننده ارائه شد. سیستم EMFAAS با پمپ کرمکی 8 کانال و شش غلطک (IPC, Ismatec, Glattbrugg-Zürich,Switzerland)، شیر دوار شش پورت (ورودی) VICI (ابزارهای والکو، هاتسون، تی ایکس، آمریکا)، 0.5 mm، لوله PTFE®، موئین سرامیکی (طول 6 سانتی متر، 0.5 میلی متر i.d.,) و کوره شعله متالیکی اتم ساز واقع در مسیر اپتیکال طیف سنج با کمک دارنده خانگی می باشد.
در شیوه آزمایشی معمولی، 500 راه حل استاندارد در جریان مانع معرفی شد(DIW) و به سلول های ذره ای یا ریزسازی در سرعت جریان 1.1mL min-1 تزریق شد. کویل نیکروم در اطراف تزریق موئین سرامیکی زخمی شد که برای گرم کردن محلول در دمای تبخیر قبل از وارد شدن به سلول استفاده شد. حرارت با تغیر ولتاژ کاربردی برای کویل بین 10 و 24 ولت کنترل شد تا از تبخیر کامل نمونه در میزان جریان متفاوت مطمئن شود.
We present an atomization system for atomic absorption spectrometry comprised by a stainless steel furnace heated by Joule effect by means of its intrinsic resistance. This new kind of furnace does not require any gases during operation. The sample is introduced with an independently controlled thermospray injector. The device outperforms conventional FAAS (Flame Atomic Absorption Spectrometry) for many analytes, giving a very safe, compact and inexpensive alternative for many analytical determinations. Full characterization of the system is presented, and theoretical simulations are contrasted with experimental data.
Introduction
Atomic Absorption Spectrometry (AAS) is one of the most widespread techniques for determination of trace elements since Alan Walsh introduced Flame Atomic Absorption Spectrometry (FAAS) in 1955.
Since then, many changes of the original idea have been investigated and several new versions of AAS have been proposed with diverse success.
The use of a flame in FAAS implies the acceptation of two drawbacks: a complex gas management system able to ensure a safe operation under every condition, and a short residence time of the sample into the optical path4 , given the fact that the flame is an open environment.
Electrothermal atomizers5-8, mainly graphite furnaces9,12 , or tungsten coils13-16 in a lower extent, became an excellent alternative to overcome the problems depicted above. Nonetheless, for these alternatives and regardless the employment of standardized temperature platform furnaces, transverse heating or matrix modification17,18, high purity argon gas flow for protection of the heated cell is mandatory.
Another strategy to confine the sample into the optical path was proposed recently by Berndt19 and refined by several groups. 20-25 This arrangement, known as thermospray flame furnace atomic absorption spectrometry (TS-FFAAS), allows the introduction of the totality of a liquid sample into a tube (or furnace) mounted over a combustion flame in the optical path of an atomic absorption spectrometer. The sample is injected via a peristaltic pump and transported to the flame furnace (FF) through a ceramic capillary directly heated by the flame, producing the so called “thermospray” (TS). The combination of the whole sample introduction and the confinement of the atomized sample has proven to optimize sensitivity for volatile elements when compared to FAAS with no need of major changes in the basic instrument.
Inspired in FFAAS and ETAAS, we present the Electrothermal Metal Furnace Atomic Absorption (EMFAAS) approach that takes advantages of both techniques. EMFAAS employs a stainless steel alloy tube as furnace. This tube of relatively high electric resistivity is very well suited to be heated by conducting a high current through its body.
Thus, the high temperature of the furnace is attained with no need of burning or protecting gases. The total liquid sample introduction is performed with the assistance of a peristaltic pump and a ceramic thermospray injector which is heated electrically and independent from the furnace heating.
In this way, EMFAAS combines electrical heating of both the FF and the TS injector, keeping away from pressurized gases either for combustion, protection or typical pneumatic nebulisation of liquid samples.
Experimental
Materials and methods
All solutions were prepared with analytical grade chemical reagents and double deionized water (DDW) obtained from a Milli-Q purification system (Millipore, Bedford, MA, USA). All glassware was washed with EXTRAN (Merck) 1% v/v and kept in 10% (v/v) HCl with further cleaning with DDW. Standard solutions of all the analytes were prepared by proper dilution of 1,000 g L-1 stock solutions (Merck Darmstadt, Germany). A digital camera Samsung NX 1100 was used to take all the temperature images. The heating of the cell was performed by means of a commercial 700 W microwave oven transformer, in which the original secondary wiring was replaced by a twoturns coil of 7 mm diameter copper multifiber cable (automotive, for batteries).
An atomic absorption spectrometer Shimadzu AA6800 (Shimadzu, Kyoto, Japan), hollow cathode lamps (Hamamatsu, Japan) and a deuterium lamp for background correction were employed throughout the measurements. Other instrumental conditions were those provided by the manufacturer.The EMFAAS system was assembled with a peristaltic pump of eight channels and six rollers (IPC, Ismatec, Glattbrugg-Zürich, Switzerland), a six-ports rotatory valve VICI (Valco Instruments, Houston, TX, USA), 0.5 mm i.d. PTFE® tubing, a ceramic capillary (0.5 mm i.d., 6 cm length) and the metallic flame furnace atomizers placed in the optical path of the spectrometer with the assistance of a homemade holder.
In a typical experimental procedure, 500 µL of standard solution was introduced in a carrier stream (DIW) and injected into the atomization cell at a flow rate of 1.1mL min-1. A Nichrome coil wounded around the ceramic injection capillary was used to heat the solution above the vaporization temperature prior to entering the cell. The heating was controlled by changing the applied voltage to the coil between 10 and 24 V to ensure full vaporization of the sample at different flow rates.
