
دانلود رایگان مقاله تنظیم هورمونی خفتگی و جوانه زنی دانه
چکیده
گیاهان دانهدار رشد می کنند تا زمانی که خفتگی در دانههای بالغ تازه حفظ شود تا زمانی که زمان مناسب برای جوانه زنی فراهم شود. خفتگی دانه و جوانه زنی دو فرآیند فیزیولوژیکی جدا هستند و انتقال از خفتگی به جوانه زنی فقط یک مرحله حیاتی در چرخه تکاملی گیاهان نیست بلکه برای تولیدات کشاورزی هم مهم است. این فرآیندها به طور دقیق توسط هورمونهای درون ریز و نشانگرهای زیست محیطی تنظیم می شوند. اگرچه ABA(آبسیزیک اسید) و Gas(ژیبرلینها) به عنوان هورمونهای گیاهی اولیه ای شناخته می شوند که به صورت آنتاگونیستی خفتگی دانه را تنظیم می کنند، یافتههای اخیر نشان می دهد که هورمون گیاهی دیگری، آکسین، برای القا و نگهداری خفتگی دانه ضروری است و بنابراین ممکن است به عنوان یک محافظت کننده کلیدی برای خفتگی دانه عمل کند. در این مقاله مروری، ما یافتههای اخیرمان را درباره شبکههای مولکولی پیشرفته خلاصه میکنیم که نقش کلیدی را در هورمونهای گیاهی دارند که خفتگی دانه و جوانه زنی را تنظیم میکند که در آن فاکتورهای رونویسی داری دومین AP-2 نقش کلیدی را ایفا میکنند. همچنین ما برهم کنش سیگنالهای هورمونی مجزایی را بررسی میکنیم که در خفتگی دانه و جوانه زنی نقش دارند و بر تعادل بین ABA/GA تمرکز میکنیم که گره اصلی را تشکیل میدهند.
مقدمه
خفتگی دانه برای زنده ماندن گیاه ضروری است و نشان می دهد که دانه جوانه می زنند فقط زمانی که شرایط محیطی بهینه باشد. بنابراین این یک صفت سازگارانه است که در گونههای گیاهی مختلفی دیده شده است و گیاهان وحشی را وادار میکند که در شرایط استرسزا در طبیعت زنده بمانند. بسیاری از دانهها از سویههای وحشی به دست آمدهاند و سطوح کاهشیافته ای از خفتگی دانه در مقایسه با خویشاوندان وحشی دیده شده است که نرخ بالاتری را بعد از شکستن ایجاد می کنند. اگرچه، فقدان یا ناکافی بودن دوره خفتگی، منجر به جوانهزنی سریع در دانههای بالغ تازه یا حتی سبز شدن دانهها قبل از انبار شدن می شود. که سبب فقدان قابل ملاحظه ای در بازده و کیفیت تولیدات کشاورزی، همچنین ایجاد مشکلاتی در مدیریت انبارداری و استفادههای صنعتی میشود.
القا، نگهداری و بعد از آن، برطرف کردن خفتگی، فرآيندهای مهم فیزیولوژیکی در دانه گیاهان میباشند. این اهمیت اکولوژیکی خفتگی دانه شامل محافظت از جوانهزنی در خارج از فصل مورد نیاز، و متعاقبا کاهش رقابت بین سویهها و حفظ حیات گیاه تحت شرایط استرسزا میباشد. به عنوان یک سوال پیچیده و چالش برانگیز در زیست شناسی، خفتگی دانه، توجهات زیادی را به خود جلب کرده است و محققان از رشتههای مختلف به آن جلب شده اند که شامل زیست شناسان گیاهی، متخصصان ژنتیک دانه، متخصصان باروری، دانشمندان علوم غذایی میباشند. اگرچه، این امر یکی از پدیدههایی در زیست شناسی گیاهی است که به میزان حداقل شناخته شده است، علیرغم اینکه فرآیند قابل توجهی است که دهه ها انجام می گرفته است. در این مقاله مروری، ما مکانیسمهای مربوط به تنظیم خفتگی دانه و جوانهزنی را خلاصه میکنیم و بر روی یافتههایی تمرکز میکنیم که شبکه کنترل کننده این حالت گذار را شامل میشوند. در بسیاری از این مطالعات، گیاه مدل Arabiopsis thaliana می باشد.
فرآیندهای مجزای خفتگی دانه و جوانهزنی
خفتگی دانه و جوانهزنی به طور گسترده در گذشته مورد مطالعه قرار گرفته است. اگرچه، آنچه خفتگی دانه را در مقیاس مولکولی مطالعه میکند، هم چنان ناشناخته مانده است. در اینجا، ما تلاش میکنیم که به این سوال پاسخ دهیم که چه دیدگاههای جدیدی بر مبنای فرآيندهای اخیر به دست آمده است.
خفتگی دانه، تعیین میکند که دانه در زمان مناسب جوانه بزند. بنابراین، در طی بلوغ، جنین باید در حالت سکون نگه داشته شود، تقریبا هیچ ذخیره سازی مواد غذایی در آن صورت نگیرد و هیچ تقسیم سلولی یا طویل شدنی را ایجاد نکند. در این حالت سکون، ژنهای افزایش دهنده جوانهزنی به طور فعالی بیان نمیشوند. بنابراین، ریشه به درون گردهها و اندوسپرم نفوذ نمیکند. در حال حاضر به خوبی شناخته شده است که ساختار کروماتین نحوه بیان ژن را تعیین میکند و بنابراین فرآيندهای چندگانه تکامل را تعیین میکند. در سالهای اخیر، بسیاری از ژنها با بازآرایی کروماتین مرتبط شده اند و برای تنظیم خفتگی دانه و جوانهزنی گزارش شدهاند. شواهد کافی نشان میدهند که ABA(آبسیزیک اسید) هم چنین در بازآرایی کروماتین نقش دارد. به عنوان مثال، ژن متیل ترانسفراز هیستونKYP/SUVH$ توسط ABA مهار میشود در حالی که استیل ترانسفراز هیستون HvGNAT/MYST القا میشود و تنظیم کنندههای اپی ژنتیکی HUB1 و RDO2 به طور شدیدی در طی القای دانه در طی تنظیم های بالادستی شرکت می کنند.
ما فرض میکنیم که بعد از فرآیند جوانه زنی، خفتگی دانه ممکن است مرتبط با ساختارهای کروماتینی ویژه ای است که در نواحی خاصی از کروموزومهای دانه وجود دارد، که در آنجا ژنهای تقویت کننده جوانه زنی حتی در حضور فاکتورهای رونویسی مرتبط هم فعال نمیشوند زیرا جایگاههای اتصال آنها به جهت پوشش فضایی، دور از دسترس میباشد. این پوشش توسط هورمونهای گیاهی انجام میشود که در این فرآیند دخالت دارند. در عوض، رهایی از خفتگی منجر به جوانه زنی، فرآيندی است که در آن ساختار کروماتینها با تیمارهای دوره سرمادهی یا تیمارهای پس از رسیدن، تغییر میکند و ژنهای در دسترس برای پیشرفت جوانه زنی را میسازد و منجر به طویل شدن سلول و تقسیم آن میشود، پوشش دانه و اندوسپرم از هم شکافته میشود و در نهایت ریشه ظهور میکند مشروط براینکه شرایط مطلوب باشد.
خفتگی در طی بلوغ دانه گسترش مییابد، درحالی که کاربرد ABA(یا حتی ABA مادری در گیاه در طی تکامل دانه) فقط جوانه زنی دانه را مهار میکند اما برای القای خفتگی دانه شکست میخورد، پس فقط ABA سنتز شده می تواند خفتگی را توسعه دهد. بنابراین، ABAهای قرار گرفته در محیطهای مختلف در بافتهای گیاهی می تواند تاثیرات متفاوتی را در خفتگی دانه یا جوانه زنی داشته باشد. به علاوه، ABI5 یک تنظیم کننده مثبت مهم در مسیر سیگنال ABA است و موتانت دارای فقدان عملکرد abi5 به مهار وابسته به ABA در جوانه زنی دانه حساس نمیباشد. اگرچه، abi5 خفتگی جایگزین در دانه را نشان نمیدهد. به علاوه، DOG1(تاخیر در جوانه زنی۱) یک ایفاگر کلیدی در القا و نگهداری خفتگی دانه است اما حساسیت ABA در dog1 بدون تغییر باقی میماند. یک مطالعه جدید نشان میدهد که DOG1 یک مکانیسم محافظت کننده پوشش خفتگی را ایجاد میکند که شامل مسیرهای وابسته به دما و ژیبرلین(GA) میباشد. مطالعات بعدی اهمیت تنظیم اپی ژنتیک برای DOG1 را نشان میدهد. دآمیلازهای هیستونی LDL1 و LDL2 خفتگی دانه را با فرآیندهای رونویسی در طی بلوغ دانه مهار میکنند. این مطالعات نشان میدهند که مسیرهای تنظیمی وابسته به DOG1 ممکن است از مسیرهای ABA یا GA مجزا باشند. این مشاهدات پیشنهاد میکند که مسیرهای سیگنالی مجزا ممکن است در تنظیم خفتگی دانه و جوانه زنی دانه سازگاری داشته باشند.
ABA و GA، دو تعیین کننده اصلی: یافته های جدید
به طور گسترده مشخص شده است که ABA و GA هورمونهای اولیهای هستند که به صورت آنتاگونیستی، خفتگی دانه و جوانه زنی را تنظیم میکنند. در طی بلوغ دانه، ABA درون ریز، درون دانه تجمع پیدا میکنند که خفتگی دانه را القا و نگهداری میکنند و بنابراین از زاد و ولد جلوگیری میکنند. در عوض، قبل از آغاز فرآيند جوانه زنی، سطح ABA درون ریز در دانه به صورت پایین دستی تنظیم میشود در حالی که محتوای GA به صورت بالادستی تنظیم میشود و این کار را با درمانهای لایه بندی و اشباعی انجام میدهند.
ABSTRACT
Seed plants have evolved to maintain the dormancy of freshly matured seeds until the appropriate time for germination. Seed dormancy and germination are distinct physiological processes, and the transition from dormancy to germination is not only a critical developmental step in the life cycle of plants but is also important for agricultural production. These processes are precisely regulated by diverse endogenous hormones and environmental cues. Although ABA (abscisic acid) and GAs (gibberellins) are known to be the primary phytohormones that antagonistically regulate seed dormancy, recent findings demonstrate that another phytohormone, auxin, is also critical for inducing and maintaining seed dormancy, and therefore might act as a key protector of seed dormancy. In this review, we summarize our current understanding of the sophisticated molecular networks involving the critical roles of phytohormones in regulating seed dormancy and germination, in which AP2-domain-containing transcription factors play key roles. We also discuss the interactions (crosstalk) of diverse hormonal signals in seed dormancy and germination, focusing on the ABA/GA balance that constitutes the central node.
INTRODUCTION
Seed dormancy is crucial to plant survival and ensures that seeds germinate only when environmental conditions are optimal. It thus is an adaptive trait in numerous seed-plant species, enabling wild plants to survive under stressful conditions in nature (Finkelstein et al., 2008). Most crops have been domesticated from wild species and show decreased levels of seed dormancy compared with their wild relatives, which ensures higher emergence rates after sowing (Lenser and Theissen, 2013; Meyer and Purugganan, 2013). However, the inappropriate loss or release of seed dormancy results in the rapid germination of freshly matured seeds or even pre-harvest sprouting (vivipary) in crops (Figure 1), causing substantial losses in yield and quality in agricultural production in addition to problems including post-harvest management and subsequent industrial utilization (Simsek et al., 2014).
Induction, maintenance, and thereafter release of seed dormancy are important physiological processes in seed plants. The ecological significance of seed dormancy includes preventing germination out of season, and consequently decreasing competition within species and ensuring plant survival under stressful conditions. As a complex and mysterious biological question, seed dormancy has attracted increasing attention from multi-disciplinary researchers, including plant biologists, crop geneticists, breeders, and food scientists. Nevertheless, it remains ‘‘one of the least understood phenomena in seed biology’’ (Finkelstein et al., 2008), despite considerable progress over past decades (Graeber et al., 2012; Rajjou et al., 2012). In this review, we summarize the mechanisms underlying the regulation of seed dormancy and germination, and focus on the emerging findings concerning the phytohormone network controlling this transition, mostly from studies with the model plant Arabidopsis thaliana.
DISTINCT PROCESSES OF SEED DORMANCY AND GERMINATION
Seed dormancy and germination has been studied intensively and extensively in the past; however, what constitutes seed dormancy at the molecular level remains largely unknown. Here, we attempt to address this question from a new viewpoint based on recent progress.
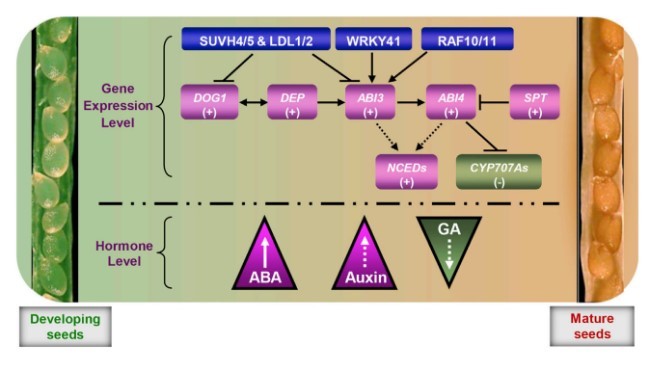
Seed dormancy ensures that seeds germinate at the appropriate time. Therefore, during maturation, the embryo must be kept in a quiescent state, mobilizing almost no stored nutrients and undergoing no cell division or elongation. In this quiescent state, germination-promoted genes are not actively expressed. Therefore, the radicle does not penetrate the testa and endosperm. It is now widely recognized that the chromatin structure determines gene expression and thereby regulates multitudinous developmental processes. In recent years, many genes associated with chromatin remodeling have been reported to regulate seed dormancy and germination (Liu et al., 2007; Saez et al., 2008; Wang et al., 2011a; Cho et al., 2012; Zheng et al., 2012). Emerging evidence shows that ABA (abscisic acid) is also involved in chromatin remodeling (Chinnusamy et al., 2008). For example, the histone methyltransferase gene KYP/SUVH4 is repressed by ABA (Zheng et al., 2012), while histone acetyltransferase HvGNAT/MYST is induced by ABA (Papaefthimiou et al., 2010), and the epigenetic regulators HUB1 and RDO2 are strikingly up-regulated during the induction of seed dormancy (Liu et al., 2011). These investigations indicated that the epigenetic regulatory-related genes possess key roles during seed maturation, which thereafter affect the seed dormancy establishment process (Figure 2).
We propose that subsequently, during the germination process, seed dormancy may be related to a characteristic chromatin structure in certain regions of chromosomes in the seed, where germination-promoted genes cannot be activated even in the presence of related transcription factors because their binding sites are unavailable due to steric hindrance, with phytohormones also involved in this process. In contrast, dormancy release leading to germination is a process in which the chromatin structure is modified by cold stratification or afterripening treatments, making the germination-promoted genes available for transcription, resulting in cell elongation and division, seed coat and endosperm rupture, and finally emergence of the radicle when conditions are favorable.
Although dormancy is established during seed maturation, whereas exogenous ABA application (or even maternal ABA in the plant during seed development) only inhibits seed germination but fails to induce seed dormancy; only ABA synthesized by the seed can establish dormancy (Kucera et al., 2005). Thus, the differently localized ABA in plant tissues possesses distinct effects on seed dormancy or germination. In addition, ABI5 is an important positive regulator in the ABA-signaling pathway, and its loss-of-function mutant abi5 is insensitive to ABA-mediated inhibition of seed germination; however, abi5 does not show altered seed dormancy (Finkelstein, 1994; Brocard-Gifford et al., 2003; Finkelstein et al., 2008). Furthermore, DOG1 (Delay of Germination 1) is a key player in the induction and maintenance of seed dormancy, but ABA sensitivity is unchanged in dog1 (Nakabayashi et al., 2012). A new study demonstrated that DOG1 mediates a conserved coat-dormancy mechanism including the temperature- and gibberellin (GA)-dependent pathways (Graeber et al., 2014). Subsequent studies suggested the importance of epigenetic regulation for DOG1. Histone demethylases LDL1 (LYSINESPECIFIC DEMETHYLASE LIKE 1) and LDL2 repress seed dormancy by regulating DOG1 (Zhao et al., 2015), and chromatin remodeling of DOG1 is also involved in dormancy cycling (Footitt et al., 2015). Furthermore, the histone methyltransferases KRYPTONITE (KYP)/SUVH4 and SUVH5 repress DOG1 and ABI3 transcription during seed maturation (Zheng et al., 2012) (Table 1). These studies demonstrated that the DOG1-mediated regulation pathway might be distinct from the ABA and/or GA pathway (Figure 2). These observations suggest that distinct signaling pathways may be adopted in the regulation of seed dormancy and seed germination.
ABA AND GA, THE MAJOR DETERMINANTS: NEWLY EMERGING EVIDENCE
It is widely recognized that ABA and GA are the primary hormones that antagonistically regulate seed dormancy and germination (Gubler et al., 2005; Finkelstein et al., 2008; Graeber et al., 2012; Hoang et al., 2014; Lee et al., 2015a). During seed maturation, endogenous ABA accumulates in the seed, inducing and maintaining seed dormancy and thus preventing vivipary (Figure 2). In contrast, before the onset of the germination process the endogenous ABA level in the seed is downregulated, while the GA content is up-regulated with imbibition and stratification treatments.
چکیده
مقدمه
فرآیندهای مجزای خفتگی دانه و جوانهزنی
ABA و GA، دو تعیین کننده اصلی: یافته های جدید
نقشهای کلیدی فاکتورهای رونویسی شامل دومینAP2 در تنظیم خفتگی دانه
اکسین: یک ایفاگر مهم جدید در خفتگی دانه
تنظیم کنندههای مختلف: سایر هورمونهای گیاهی دخیل در خفتگی دانه و جوانهزنی
دیدگاه ها و نکات نتیجه گیری
منابع
ABSTRACT
INTRODUCTION
DISTINCT PROCESSES OF SEED DORMANCY AND GERMINATION
ABA AND GA, THE MAJOR DETERMINANTS: NEWLY EMERGING EVIDENCE
KEY ROLES OF AP2 DOMAINCONTAINING TRANSCRIPTION FACTOR IN SEED DORMANCY REGULATION
AUXIN: A NEW MASTER PLAYER IN SEED DORMANCY
DIVERSE REGULATORS: OTHER PHYTOHORMONES INVOLVED IN SEED DORMANCY AND GERMINATION
CONCLUDING REMARKS AND PERSPECTIVES
REFERENCES
