
دانلود رایگان مقاله اکولوژی و مدیریت شب پره پشت الماسی
چکیده
رشد چشمگیر کشاورزی و تولید بیشتر محصولات نباتی و دانه های روغنی طی دو دهه گذشته، وضعیت آفات شب پره پشت الماسی (DBM)، Plutella xylostella L. را افزایش داده است و در حال حاضر برآورد شده است که سالانه 4 تا 5 میلیارد دلار برای اقتصاد جهانی هزینه در بر داشته باشد. درک ما از برخی از جنبه های اساسی بیولوژی و اکولوژی DBM، به ویژه در مورد روابط گیاهان میزبان، تعاملات تریتروفیک و مهاجرت به طور قابل توجهی بهبود یافته است، اما دانش ما از جنبه های دیگر مانند توزیع جهانی و فراوانی نسبی آن، به طرز شگفت آوری محدود باقی مانده است. کنترل بیولوژیکی هنوز هم تقریباً به طور انحصاری روی تعدادی از انواع پارازیتوئیدهای (انگلهای) هیمانوپتران متمرکز است. اگرچه اینها می توانند بسیار مؤثر باشند ، اما حشره کش ها همچنان اساس مدیریت را تشکیل می دهند؛ استفاده نامناسب از آنها پارازیتوئیدها را مختل می کند و باعث مقاومت در برابر محصولات موجود می شود. درک اکولوژیکی بهبود یافته و در دسترس بودن مجموعه ای از حشره کش های انتخابی بسیار مؤثر در طول دهه 1990، پایه و اساس رویکردهای مدیریت یکپارچه آفات پایدار و از نظر اقتصادی مقاوم (IPM) را فراهم کرد. با این حال، رجوع مکرر به برنامه های حشره کش برنامه ریزی شده منجر به مقاومت در برابر این ترکیبات اخیراً معرفی شده و خرابی برنامه های IPM شده است. در حال حاضر، فناوری های اثبات شده ای برای مدیریت پایدار DBM وجود دارند، اما غلبه بر موانع اتخاذ پایدار آنها همچنان به عنوان یک چالش بزرگ باقی مانده است.
مقدمه
هنوز بیست سال نشده است که Talekar و Shelton (168)، بررسی های برجسته خود در مورد شب پره پشت الماسی (DBM)، Plutella xylostella L. (Lepidoptera: Plutellidae) را منتشر کرده اند، در حال حاضر به طور قابل توجهی در مورد جنبه های بسیار زیادی از زیست شناسی (34، 147، 155، 161)، بوم شناسی (125، 129) و ژنتیک (12) آن آگاهی داریم. با وجود این پیشرفت ها، DBM وضعیت خود به عنوان مخرب ترین عضو مجتمع آفات حشرات که به مزارع نباتی کلزا (Brassica) در مناطق مختلف جهان حمله می کنند (34، 147، 155، 161، 165، 167) را حفظ کرده است و در صورت تناوب این حملات، به طور فزاینده یک تهدید مهم برای تولید کلزا محسوب خواهد شد (45).
بین سال های 1993 و 2009، در سطح جهانی محصولات نباتی Brassica تا 39 درصد افزایش یافته است و در سال 2009 تخمین زده شده است که 3.4 میلیون هکتار در سراسر جهان (38) رشد داشته باشد. همزمان با این تغییر، شیوه های کشاورزی افزایش یافت و به دنبال آن محصول کلم نیز 27 درصد رشد داشته اس و اکنون نباتات برزیکا بیش از 26 میلیارد دلار برای اقتصاد جهانی کمک کرده است (38). در همان دوره، سطح زیر کشت گیاهان روغنی کاشته شده 59٪ افزایش یافت، و در سال 2009 بیش از 31 میلیون هکتار در سطح جهان کشت شد (38)، و این غالباً در مناطقی بود که قبلاً در آنجا این محصول کشت نشده بود. این تغییرات منجر به تغییرات قابل توجهی در بسیاری از مناظر محلی شده است (45) و چالش های تجدید نظر شده ای را برای مدیریت DBM (143) فراهم اورده است.
Talekar و Shelton (168) بر اهمیت اقدامات کارگاه های بین المللی در مورد DBM که در سالهای 1985 و 1990 برگزار شد (165 ، 167)، تأکید کردند. چهار کارگاه اضافی که بین سالهای 1996 و 2011 تشکیل شد و هر یک اقدامات ارزشمندی درمورد تحقیقات معاصر DBM انجام دادند (34، 147، 155، 161). با توجه به این بدنه کار، از بسیاری از نشریات موجود در مقالات دانشگاهی و از تغییراتی که از سال 1993 در تولید محصولات زراعی برزیکا رخ داده است، موقع آن رسیده است که دانش خود در مورد اکولوژی و مدیریت DBM را مورد بررسی قرار داده و آن را در متن فعلی قرار دهیم.
توزیع DBM، مدیریت فعلی و هزینه های اقتصادی
علیرغم وضعیت آفات DBM و ادعاهایی در مورد اینکه این گسترده ترین توزیع از تمام پولک بالان (Lepidoptera) است (168)، درک فعلی ما از توزیع جهانی و فراوانی نسبی آن محدود است (187). نقشه توزیع اصلی (25) ترکیبی از سوابق توزیع ناقص است ، و این نسخه اخیراً توسط نسخه ای جایگزین شده است که کشورهایی را که گزارش DBM در آن گزارش شده است را ثبت می کند (16).
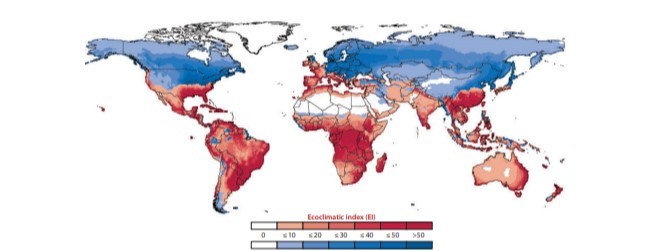
Zalucki و Furlong (187) یک مدل بیوشیمیایی را برای DBM توسعه داده و مورد تأیید قرار دادند که توزیع اصلی آن را پیش بینی می کند که این در طول سال و همچنین مناطقی که می تواند آفت فصلی باشد، ادامه می یابد (شکل 1). این مدل همچنین فنولوژی (پدیده شناسی) فصلی متغیر نشان داده شده توسط DBM در سراسر دامنه وسیع مربوط به آن را پیش بینی می نماید (189) و تأثیرات محدودکننده جمعیت باران زیاد و دمای شدید (82) که می تواند از مقاومت در طول سال جلوگیری کند را نشان می دهد (55، 58). علاوه بر این، این مناطقی را مشخص می کند که از نظر فصلی برای رشد جمعیت DBM مناسب هستند، مناطقی که در آن شیوع آفات می تواند به دنبال هجوم DBM افزایش یابد (31).
با وجود تلاش قابل توجه برای توسعه رویکردهای یکپارچه برای مدیریت DBM (39، 51)، اکثریت قریب به اتفاق محصولات برزیکا به صورت پیشگیری با حشره کش ها می باشند (51). این شیوه در کشورهای گرمسیری بسیار کاربرد دارد جاهایی که محصولات غیر منتخب، اغلب مخلوط می شوند، و این شیوه معمولاً 1 تا 2 بار در هفته بر روی آنها اعمال می شود (5، 51، 96)؛ با این حال، استفاده بیش از حد از حشره کش ها در برابر DBM محدود به جهان در حال توسعه یا مناطق استوایی نمی شود (95). چنین شیوه هایی باعث افزایش انتخاب مقاومت حشره کش (81)، نابود کردن دشمنان طبیعی (44) و آلوده کردن محیط می شود. آنها همچنین گران قیمت هستند، و یک مطالعه جدید (189) تخمین زده است که كنترل سالانه DBM در محصولات گیاهی برزیکا به تنهایی 1.4 میلیارد دلار در سراسر جهان هزینه در بر دارد، در صورت ضرر در بازده به 2.7 میلیارد دلار افزایش می یابد و در صورت ضرر و زیان DBM و هزینه های كنترل به 4-5 میلیارد دلار نیز می رسد تا صنعت کلزا در سراسر جهان افزایش یابد.
بیولوژی و اکولوژی DBM: پیشرفت ها از سال 1993
دشمنان طبیعی DBM
طیف گسترده ای از دشمنان طبیعی، از جمله پارازیتوئیدها، بندپایان غارتگر، ویروس ها، میکروسپوریدیا، قارچ های بیماری زا و باکتری ها به DBM حمله می کنند. تحقیقات اساسی و کاربردی قابل توجهی در طی دو دهه گذشته روی این ارگانیسم ها به ویژه در برنامه های کنترل بیولوژیکی کلاسیک با استفاده از انگلهای هیمانوپتران (129) متمرکز شده است.
پارازیتوئیدهای هایمانوپتران. کنترل بیولوژیکی کلاسیک برای DBM از سال 1936 آغاز شد، زمانی که پارازیتوئید لارو -شفیره Diadegma semiclausum (Hellen) (پرده بالان: Ichneumonidae) و پارازیتوئید مگس Diadromus collaris (Gravenhorst) (پرده بالان: Ichneumonidae) با موفقیت در نیوزیلند از انگلستان (168) معرفی شد و منجر به معرفی بیشتر هر دو گونه از نیوزلند به اندونزی (180) ، استرالیا (186) و مالزی (104) شد. به دنبال واردات D. semiclausum از اندونزی، پارازیتوئید در تایوان (169) مستقر شد و مواد اولیه لازم را برای معرفی موفقیت آمیز بعدی در فیلیپین (177) ، هند (21) ، لائوس ، ویتنام ، چین (166) و کنیا فراهم کرد. در سال 2005، D. semiclausum از مالزی با موفقیت در مناطق مرتفع تایلند (174) مستقر شد. از این رو، منشاء جمعیت D. semiclausum که در سراسر آسیا، استرالیا و بخش هایی از آفریقا مستقر شد، کاملاً واضح و مستند است و در نهایت از انگلستان سرچشمه می گیرد. به طور مشابه، زنبور پارازیتوئید Cotesia vestalis (=plutellae) هالیدی (پرده بالان: Braconidae) بیش از 20 مقدمه کنترل بیولوژیکی کلاسیک را به خود اختصاص داده است که بسیاری از آنها موفق بوده اند (28، 166). علاوه بر این، به نظر می رسد که این پارازیتوئید دارای توزیع طبیعی وسیع تری نسبت به D. semiclausum است و برای حمله به DBM در بسیاری از مناطق بدون هیچ سوابق مقدماتی ثبت شده است [به عنوان مثال، مالزی (104)، تایوان (166)، ویتنام (166)، چین (87) و ژاپن (3)]. پارازیتوئید لارو-شفیره Oomyzus sokolowksii Kurdjumov (پرده بالان: Eulophidae) نیز در کشورهایی که بدون سابقه مقدماتی ثبت شده است [به عنوان مثال: استرالیا (46)، ژاپن (166)، آفریقای جنوبی (71)، و کره شمالی (39)]؛ با این حال، به عمد وارد چین و تایوان شده است (166). به طور مشابه، پارازیتوئید های شفیره D. collaris و Diadromus subtilicornis (گریون هرست) (پرده بالان: Ichneumonidae) توزیع طبیعی گسترده ای دارند (28). از زمان انتشار اولیه D. collaris در نیوزلند، این در بسیاری از کشورها معرفی شده است (28)، اما D. sub- tilicornis هرگز برای کنترل بیولوژیکی معرفی نشده است (28).
Abstract
Agricultural intensification and greater production of Brassica vegetable and oilseed crops over the past two decades have increased the pest status of the diamondback moth (DBM), Plutella xylostella L., and it is now estimated to cost the world economy US$4–5 billion annually. Our understanding of some fundamental aspects of DBM biology and ecology, particularly host plant relationships, tritrophic interactions, and migration, has improved considerably but knowledge of other aspects, e.g., its global distribution and relative abundance, remains surprisingly limited. Biological control still focuses almost exclusively on a few species of hymenopteran parasitoids. Although these can be remarkably effective, insecticides continue to form the basis of management; their inappropriate use disrupts parasitoids and has resulted in field resistance to all available products. Improved ecological understanding and the availability of a series of highly effective selective insecticides throughout the 1990s provided the basis for sustainable and economically viable integrated pest management (IPM) approaches. However, repeated reversion to scheduled insecticide applications has resulted in resistance to these and more recently introduced compounds and the breakdown of IPM programs. Proven technologies for the sustainable management of DBM currently exist, but overcoming the barriers to their sustained adoption remains an enormous challenge.
INTRODUCTION
Twenty years have passed since Talekar & Shelton (168) published their landmark review of the diamondback moth (DBM), Plutella xylostella L. (Lepidoptera: Plutellidae), and we now know considerably more about many aspects of its biology (34, 147, 155, 161), ecology (125, 129), and genetics (12). Despite these advances, DBM has retained its status as the most destructive member of the different insect pest complexes that attack Brassica vegetable crops in various parts of the world (34, 147, 155, 161, 165, 167), and it is increasingly considered a significant, if sporadic, threat to canola production (45).
Between 1993 and 2009 the global area of Brassica vegetable crops increased by 39%, and in 2009 an estimated 3.4 million hectares were grown worldwide (38). Concomitant with this change was an intensification of farming practices, with cabbage yields increasing by 27%, and Brassica vegetables now contribute more than US$26 billion to the world economy (38). Over the same period, the area of oilseed rape planted increased by 59%, and in 2009 more than 31 million hectares were cultivated worldwide (38), often in regions where the crop was not previously grown. These changes have resulted in considerable modifications to many local landscapes (45) and provide revised challenges for DBM management (143).
Talekar & Shelton (168) stressed the importance of the proceedings of the international workshops on DBM held in 1985 and 1990 (165, 167). An additional four workshops convened between 1996 and 2011 and each published valuable proceedings on contemporary DBM research (34, 147, 155, 161). In consideration of this body of work, of the many publications in the academic literature, and of the changes that have occurred in Brassica crop production since 1993, it is timely to review our knowledge of DBM ecology and management and to set it in the current context.
DBM DISTRIBUTION, CURRENT MANAGEMENT, AND ECONOMIC COSTS
Despite the pest status of DBM and assertions that it has the most extensive distribution of all Lepidoptera (168), current understanding of its global distribution and relative abundance is limited (187). The original distribution map (25) is a composite of incomplete distribution records, and this has recently been superseded by a version that simply records countries where DBM has been reported (16).
Zalucki & Furlong (187) developed and validated a bioclimatic model for DBM that predicts its core distribution, where it persists year-round, as well as regions where it can be a seasonal pest (Figure 1). The model also predicts the variable seasonal phenologies exhibited by DBM across its vast range (189), illustrating the population-limiting effects of high rainfall and extreme temperatures (82) that can preclude year-round persistence (55, 58). Further, it identifies regions that are seasonally suitable for DBM population growth, where pest outbreaks can be promoted following DBM influxes (31).
Despite the considerable effort to develop integrated approaches to DBM management (39, 51), the vast majority of Brassica crops are treated prophylactically with insecticides (51). This practice is most acute in tropical countries, where nonselective products, which are often mixed, are typically applied 1 to 2 times per week (5, 51, 96); however, excessive use of insecticides against DBM is not restricted to the developing world or the tropics (95). Such practices promote the selection for insecticide resistance (81), destroy natural enemies (44), and contaminate the environment. They are also expensive, and a recent study (189) estimated that annual DBM control in Brassica vegetable crops alone costs US$1.4 billion worldwide, rising to US$2.7 billion if yield losses are included and to $4–5 billion if DBM losses and control costs to the worldwide canola industry are added.
DBM BIOLOGY AND ECOLOGY: ADVANCES SINCE 1993
Natural Enemies of DBM
A wide range of natural enemies, including parasitoids, arthropod predators, viruses, microsporidia, pathogenic fungi, and bacteria, attack DBM. Considerable basic and applied research has focused on these organisms over the past two decades, particularly on classical biological control programs utilizing hymenopteran parasitoids (129).
Hymenopteran parasitoids. Classical biological control for DBM began in 1936 when the larvalpupal parasitoid Diadegma semiclausum (Hellen) (Hymenoptera: Ichneumonidae) and the pupal ´ parasitoid Diadromus collaris (Gravenhorst) (Hymenoptera: Ichneumonidae) were successfully introduced into New Zealand from the United Kingdom (168), leading to further introductions of both species from New Zealand into Indonesia (180), Australia (186), and Malaysia (104). Following importation of D. semiclausum from Indonesia, the parasitoid was established in Taiwan (169), providing stock material for subsequent successful introductions into the Philippines (177), India (21), Laos, Vietnam, China (166), and Kenya (89). In 2005, D. semiclausum from Malaysia successfully established in the highland regions of Thailand (174). Hence, the provenance of the D. semiclausum populations that have established throughout Asia, Australasia, and parts of Africa is clear and well documented and all ultimately originate from the United Kingdom. Similarly, the larval parasitoid Cotesia vestalis (=plutellae) Haliday (Hymenoptera: Braconidae) has been the subject of more than 20 classical biological control introductions, and many of them have been successful (28, 166). In addition, this parasitoid appears to have a wider natural distribution than D. semiclausum, and it has been recorded to attack DBM in many regions [e.g., Malaysia (104), Taiwan (166), Vietnam (166), China (87), and Japan (3)] with no records of introductions. The larval-pupal parasitoid Oomyzus sokolowksii Kurdjumov (Hymenoptera: Eulophidae) has also been recorded in countries with no records of introduction [e.g., Australia (46), Japan (166), South Africa (71), and North Korea (39)]; however it has been deliberately introduced into China and Taiwan (166). Similarly, the DBM pupal parasitoids D. collaris and Diadromus subtilicornis (Gravenhorst) (Hymenoptera: Ichneumonidae) have widespread natural distributions (28). Since the initial release of D. collaris in New Zealand, it has been introduced into many countries (28), but D. subtilicornis has never been introduced for biological control (28).
چکیده
مقدمه
توزیع DBM، مدیریت فعلی و هزینه های اقتصادی
بیولوژی و اکولوژی DBM: پیشرفت ها از سال 1993
دشمنان طبیعی DBM
تعاملات گیاه میزبان DBM
جنبش محلی و مهاجرت
DBM در مقیاس دور نما
مشخصات ، مقاومت و استراتژي هاي مديريت مقاومت يكپارچه
مقاومت در برابر حشره کش و DBM
مکانیسم های مقاومت و مقاومت متقاطع و مقاومت چندگانه در DBM
مدیریت مقاومت در برابر حشره کش ها: نظریه و پیاده سازی
فناوری های جایگزین و اضطراری و استراتژی های مدیریت
محصولات زراعی تراریخته (تراژنی) Bt برزیکا
تکنیک های کنترل ژنتیک
دامهای(تله) زراعی و دستکاری مقاومت گیاه میزبان
مدیریت تلفیقی آفات: محدودیت های اجرایی و پذیرش (اتخاذ)
نتیجه گیری
منابع
Abstract
INTRODUCTION
DBM DISTRIBUTION, CURRENT MANAGEMENT, AND ECONOMIC COSTS
DBM BIOLOGY AND ECOLOGY: ADVANCES SINCE 1993
Natural Enemies of DBM
DBM–Host Plant Interactions
Local Movement and Migration
DBM at the Landscape Scale
INSECTICIDES, RESISTANCE, AND INTEGRATED RESISTANCE MANAGEMENT STRATEGIES
Insecticide Resistance and DBM
Resistance Mechanisms and Cross-Resistance and Multiple Resistance in DBM
Insecticide Resistance Management: Theory and Implementation
ALTERNATIVE AND EMERGENT TECHNOLOGIES AND MANAGEMENT STRATEGIES
Transgenic Bt-Brassica Crops
Genetic Control Techniques
Trap Crops and the Manipulation of Host Plant Resistance
INTEGRATED PEST MANAGEMENT: CONSTRAINTS TO IMPLEMENTATION AND ADOPTION
SYNTHESIS AND CONCLUSIONS
LITERATURE CITED
