
دانلود رایگان مقاله اثرات اتوکلاو و پروتئین ضد میکروبی در فعالیت های سماق ایرانی
چکیده
اگر چه بسیاری از ترکیبات در حال حاضر برای استفاده در غذایی به عنوان ضد میکروب تایید شده اند، تحقیق برای پیدا کردن تعداد بیشتری از این ترکیبات هنوز هم مدنظر است زیرا در حال حاضر بیشتر ضد میکروب های غذایی تایید شده دارای کاربردهای محدود ناشی از فعل و انفعالات ترکیب غذایی هستند. یافتن ضد میکروب های جدید غذایی نیاز به بررسی های پرهزینه دارد. عوامل سنتی گیاهی ضد میکروبی مانند سماق می توانند نقش مهمی را بازی نمایند. اگر رواج سماق در مرتبه عامل ضد میکروبی غذایی مورد تقاضا باشد ، ثبات و تعاملات آن ارزیابی شود. ارزیابی ثبات حرارتی سماق با اندازه گیری MICها و MBCها از عصاره در برابر برخی از این باکتریهای مرتبط با غذایی در وضعیت فوق العاده اتوکلاو انجام شده است. اجزای اصلی عصاره سماق ترکیبات جوهر مازو هستند.. جوهر مازو دارای تعامل با نمک و پروتئین است، و آنها در چنین شرایطی ته نشین می شوند. فعالیتهای ضد میکروبی عصاره با روش انتشار در حضور نمک و پروتئین اندازه گیری شده و با عصاره خالص مقایسه شده است. نتایج نشان می دهد که سماق پایدار درمقابل حرارت است اما دارای تعاملاتی با نمک و پروتئین ها است که باعث کاهش فعالیت خود در مقابل باکتری های gram منفی می شود.
مقدمه
یکی از پیشرفت های مهمی در تاریخچه انسان، قابلیت حفظ غذایی و مهار فساد غذایی توسط تکنیک های حفظ، یعنی ضد میکروبهای غذایی است. ضد میکروبها غذایی، ترکیبات افزوده شده یا موجود در غذا هستندکه که باعث تاخیر در رشد میکروبی و یا کشتن میکرو ارگانیسم ها می شوند. اگر چه بسیاری از ترکیبات در حال حاضر برای استفاده در غذایی به عنوان ضد میکروبها مورد تایید هستند، تحقیقات برای پیدا کردن تعداد بیشتری از این ترکیبات هنوز مدنظر است زیرا بسیاری از ضد میکروبها غذایی سنتی، در حال حاضر مورد تایید دارای کاربردهای محدود ناشی از تعاملات ترکیب غذایی هستند. تعامل با اجزای غذایی باعث کمتر در دسترس بودن ضد میکروبهای غذایی برای مهار میکروارگانیسمها محصولات غذایی می شود و یک عامل ضد میکروبی غذایی خوب باید حداقل چنین فعل و انفعالات باشد. ضمناً باید غیر سمی، غیر آلرژی زا ، ارزان و پایدار در برابر فرآیندهایی باشد که در معرض آن است [1]. پیدا کردن ضد میکروبی غذایی نگهدارنده جدید با چنین ویژگی هایی نیاز به صرف وقت بسیار و هزینه زیاد دارد. به عنوان مثال، عامل ضد میکروبی غذایی جدید باید عبور به طور کلی از آزمایشات سم شناسی سخت تایید شده توسط سازمان های نظارتی بین المللی عبور نماید.
مطالعات اخیر نشان داد که ضد میکروبهای گیاهی می تواند نقش مهمی بازی کنند. از آنجا که یکی از راه های نشان دادن غیر سمی بودن ضد میکروبها مصرف مداوم آنها به عنوان یک ماده غذایی در طی یک دوره طولانی است، ادویه جات می توانند کاندیداهای خوبی برای تحقیق در مورد ضد میکروبهای غذایی باشند [1]. سماق یک ادویه ایرانی است که بسیار در غذاهای ایرانی استفاده می شود. فعالیت های ضد میکروبی این ادویه در مطالعات قبلی ما گزارش شده است [2 ، 3]. اما اگر تقاضا به ارتقاء سماق به مرتبه عامل ضد میکروبی غذایی صورت گیرد، ثبات و تعاملات آن باید ارزیابی شود. این مطالعه سعی در ارزیابی پایداری اثرات ضد میکروبی سماق در طول اتوکلاو دارد. همچنین با توجه به این مسئله که بسیاری از محصولات غذایی دارای میزان کمتر یا بیشتر نمک و پروتئین هستند، این تحقیق به دنبال تعاملات ممکن از این اجزای سازنده با سماق است.
مواد و روشها
میکروارگانیسم ها و شرایط رشد
دو استاندارد gram-منفی و gram-مثبت و یک باکتری بالینی جدا شده مرتبط با مواد غذایی در آزمایشات مورد استفاده قرار گرفته است. Staphylococcus aureus 6539-P, Escherichia coli ATCC 8739, and Pseudomonas aeruginosa ATCC 9027 در گروه کنترل دارویی و مواد غذایی ، دانشکده داروسازی ، علوم پزشکی / دانشگاه تهران وجود دارند در حالی که Salmonella ty یک کشش بالینی جدا شده و شناسایی شده در آزمایشگاه کنترل میکروبیولوژیکی این بخش بود. کشت میکروب در ازمایشگاه مقدار باکتری در 20 ٪ گلیسرول پی بی اس (بافر فسفات سالین) در -70 درجه سانتی گراد نگهداری شدند. کشت فعال میکروب در ازمایشگاه توسط مایه کوبی 100 میکرولیتر از تعلیق موجودی میکروبی ذوب شده به 5 میلی لیتر از مواد مغذی (merck ، آلمان) توسط رشد نهفته شبانه در 37 درجه سانتی گراد تولید شد. کشت هماهنگ باکتریایی در ازمایشگاه با انتقال 100 میکرولیتر از سلول های گیاهی به Muller Hinton broth رشد نهفته به مدت 24 ساعت و در37 درجه سانتیگراد آماده سازی شد. سلول ها با سانتریفوژ در 1600g به مدت 10 دقیقه ، با PBS شسته شده، چرخش دوباره در 1600g و رقیق شده در آب مقطر برای دست آوردن cfu/ml برآورد شده برداشت شدند [4].
آماده سازی نمونه
سماق ایرانی است که در پوسته خارجی Rhus coriaria L وجود دارد در بازار داخلی به دو صورت قهوه ای (رسیده) قرمز و قهوه ای (نیمه رسیده) قابل دسترسی می باشد. سماق رسیده قهوه ای مورد استفاده در این مطالعه خریداری شده از بازار گیاه شناسی محلی است و توسط گیاهدان دانشکده داروسازی دانشگاه علوم پزشکی تهران (TEH)از مجموعه گیاهان خشک شناسایی شده است. عصاره آب نمونه با استفاده از روش لاغری با استفاده از آب استریل40 درجه سانتی گراد آماده شده است. عصاره در تبخیرکننده خلاء دوار قرار گرفته است (Buchi, Switzerland) و با رشد نهفته در 40 درجه خشک شده است.
تعیین ثبات در اتوکلاو
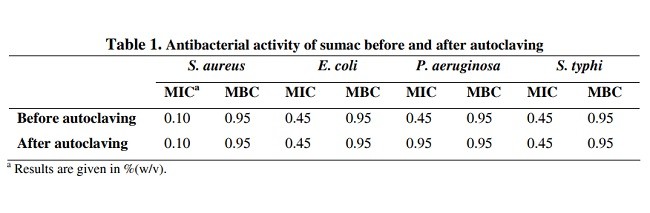
500 میلی گرم عصاره خشک سماق آب مقطر استریل در غلظت 50 درصد رقیق شده است (w/v). عصاره تقسیم به دو بخش شد. یک بخش فیلتر شده استریل و دیگری در وضعیت اتوکلاو (121 درجه و 1 اتمسفر) برای 15 دقیقه قرار گرفت. برای تعیین حداقل غلظت مهاری (MIC)، رقت نمونه های بین 0.03 تا 3.75 درصد در Muller Hinton broth آماده شدند. غلظت نهایی باکتریهای موجود در لوله های منفرد cfu/ml بود. لوله های کنترل حاوی هیچ عصاره ای نبودند. پس از رشد نهفته در 37 درجه سانتی گراد، لوله های آزمایش برای رشد ممکن مورد بررسی قرار گرفتند و MIC هر قسمت از عصاره به صورت کمترین غلظت تعیین شد که با هیچ رشدی پایان نمی یافت. لوله های حاوی غلظت بالا، MICها در صفحات Muller hinton برای به دست اوزدن مینیمم غلظت های باکتریایی (MBC) برای نمونه های منفرد در مقابل کشش های آزمایش شده گسترده می شدند.
تعیین اثر متقابل ممکن با نمک و ژلاتین
اجزای اصلی عصاره سماق ترکیبات جوهر مازو هستند. جوهر مازو دارای فعل و انفعالات با نمک و پروتئین است [5]. به منظور ارزشیابی اثار این فعل و انفعالات در فعالیت های ضد میکروبی عصاره، 1 میلی گرم از عصاره سماق (20 درصد) به 2 میلی گرم محلول ژلاتین (1 درصد) یا محلول سالین (10 درصد) به طور جداگانه اضافه شد و نتیجتاً به طور تقریبی تمام ترکیبات جوهر مازوی عصاره به شکل رسوب جدا شدند. بعد از سانتریفیوژ در 10 دقیقه در 5000g، 100 میکرولیتر جوهر مازو شفاف شناور بر روی اب با روش انتشار در ازای Staphylococcus aureus and Pseudomonas aeruginosa آزمایش شد. بعد از رشد نهفته در شب در 37 درجه، نواحی ممانعت حول چاه ها بر حسب میلی متر با استفاده از یک ک.لیس اندازه گیری شدند.
Abstract
Although many compounds have already approved for use in food as antimicrobials, research for finding greater number of these compounds are still interesting because most currently approved food antimicrobials have limited applications due to food compound interactions. Finding new food antimicrobials requires expensive investigations. Traditional herbal antimicrobial agents like sumac can play an important role. If it is demanded to promote sumac to the rank of a food antimicrobial agent, its stability and interactions must be evaluated. The evaluation of sumac heat stability was done by measuring MICs and MBCs of the extract against some food- related bacteria in extreme condition of autoclaving. The main components of sumac extract are tannic compounds. Tannins have interactions with salt and proteins, and they become sediment in such conditions. Antimicrobial activities of the extract were measured by well diffusion method in the presence of salt and protein and compared with the activities of pure extract. The results show that sumac is heat stable but has interactions with salt and proteins that reduce its activity against Gram- negatives.
Introduction
One of the major advances in human history is ability to preserve food and inhibit food spoilage by preservation techniques, namely food antimicrobials. Food antimicrobials are compounds added to or present in foods that retard microbial growth or kill microorganisms. Although many compounds have already approved for use in food as antimicrobials, research for finding greater number of these compounds are still interesting because most of the traditional, currently approved food antimicrobials have limited applications due to food compound interactions. Interaction with food components makes food antimicrobials less available to inhibit microorganisms of food products and a good food antimicrobial agent should have least such interactions. It should be also nontoxic, non- allergenic, cheap, and stable to any processes to which it is exposed [1]. Finding new antimicrobial food preservative with such characteristics requires many time consuming and expensive investigations. For instance, a novel food antimicrobial agent should pass generally very strict toxicological tests to be approved by international regulatory agencies.
Recent studies showed that herbal antimicrobials can play an important role. Because one way of indicating non- toxicity of antimicrobials is their continuous consumption as a food over a long period, spices can be good candidates for investigating food antimicrobials [1]. Sumac is an Iranian spice used vastly in Iranian cuisine. Antimicrobial activities of this spice were reported in our previous studies [2, 3]. But if it is demanded to promote sumac to the rank of a food antimicrobial agent, its stability and interactions must be evaluated.This study tries to assess stability of antimicrobial effects of sumac during autoclaving. Also, regarding this issue that most of the food products have -more or lesssome amounts of salt or protein, this research seeks possible interactions of these components with sumac.
Materials and methods
Microorganisms and growth conditions
Two Gram-negative and one Grampositive standard and one clinical isolated food-related bacteria were used in the experiments. Staphylococcus aureus 6539-P, Escherichia coli ATCC 8739, and Pseudomonas aeruginosa ATCC 9027 were stocked in the Department of pharmaceutical and Food Control, School of Pharmacy, Medical Sciences/ University of Tehran while Salmonella typhi was a clinical strain isolated and identified in the microbiological control laboratory of the department. Stock cultures of bacteria were kept in 20% glycerol PBS (phosphate buffered saline) at −70 °C. Active cultures were generated by inoculating 100 µl of the thawed microbial stock suspensions into 5 ml nutrient broth (Merck, Germany) followed by overnight incubation at 37 °C. Freshly synchronized cultures of bacterial strains were prepared by successively transferring 100 µl of the vegetative cells into Muller Hinton broth and incubating for 24 h at 37 °C. The cells were harvested by centrifugation at 1600g for 10 min, washed with PBS, spun at 1600g again and diluted in sterile water to obtain 108 cfu/ml as estimated by the surface plate counting method [4].
Sample preparation
Iranian sumac which is the ground unsalted epicarps of Rhus coriaria L. is available in the local market in two forms of brown (ripened) and brown red (semi ripened). The brown ripened sumac used in this study was purchased from the local botanical market and were identified by the Herbarium of the School of Pharmacy (TEH), Tehran University of Medical Sciences. Water extracts of the specimen was prepared by maceration method using 40°C sterile distilled water. Extract was concentrated in a vacuum rotary evaporator (Buchi, Switzerland) and were dried by incubation in 40°C.
Stability determination during autoclaving
500 mg of the sumac dry extract was diluted in sterile distilled water to make a concentration of 50% (w/v). The extract was divided into two parts. One part was filter sterilized and the other was exposed to autoclaving condition (121°C and 1 atm) for 15 minutes. To determine the minimum inhibitory concentration (MIC), serial dilutions of the samples were prepared between 0.03 to 3.75% (w/v) in Muller Hinton broth. Final concentration of bacteria in individual tubes was 106 cfu/ml. Control tubes contained no extract. After overnight incubation at 37°C, the test tubes were examined for possible growth and MIC of each part of the extract was determined as the lowest concentration that ended with no growth. Tubes containing concentrations above the MICs were streaked onto Muller Hinton agar plates to achieve minimum bactericidal concentrations (MBC) of individual samples against the tested strains.
Determination of possible interactions with salt and gelatin
The main components of sumac extract are tannic compounds. Tannins have interactions with salt and proteins [5]. To evaluate the effects of these interactions in antimicrobial activities of the extract, 1 ml of sumac extract (20%) was added to 2 ml of a gelatin solution (1%) or a saline solution (10%) separately and consequently, nearly all of the tannic compounds of the extract were separated in the form of sediment. After centrifuging for 10 minutes at 5000g, 100 µl of the transparent and tannin-less supernatant was examined against Staphylococcus aureus and Pseudomonas aeruginosa by well diffusion method. After overnight incubation at 37°C, the inhibition zones around wells were measured in millimeter using a caliper.
چکیده
مقدمه
مواد و روشها
میکروارگانیسم ها و شرایط رشد
آماده سازی نمونه
تعیین ثبات در اتوکلاو
تعیین اثر متقابل ممکن با نمک و ژلاتین
نتایج
بررسی
Abstract
Introduction
Materials and methods
Microorganisms and growth conditions
Sample preparation
Stability determination during autoclaving
Determination of possible interactions with salt and gelatin
Results
Discussion
