
دانلود رایگان مقاله اندازهگیری راکتیوتیه عروقی برای غلبه بر جداشدگی عصبی عروقی در تصویربرداری
چکیده
هدف: MRI عملکردی (fMRI) قبل از عمل جراحی با پاسخ خاموش BOLD ناشی از راکتیویته عروقی غیرنرمال و جداشدگی عصبی عروقی مجاور تومورهای بدخیم مغزی محدوده شده است. با استفاده از حبس کردن نفس و ادغام این دادهها در تحلیل BOLD رفع این محدودیت و شناسایی دقیقتر مناطق سخنوری مجاور تومورهای مغزی با ارزیابی و بررسی مستقل راکتیویته عروقی پیشنهاد میکنیم.
روشکار: راکتیویته موضعی عروقی با استفاده از الگوی حبس کردن تنفس با زمان یکسان به عنوان موتور عملکردی و وظایف زبانی در 16 نفر از بیماران (9 بیمار گلیوبلاستوما،1 آستروسیتومای اناپلاستیک، 5 مورد آستروسیتومای درجه پایین و 1 بیمار متاستاز) مشخص شد. یک مدل مبتنی بر انسجام را برای تحلیل BOLD fMRI ایجاد کردیم که همودینامیکهای تغییر یافته در مجاورت تومورهای مغزی را در نظر گرفته است.
یافتهها: نقشههای فعالسازی با استفاده از مدل انسجام بدست آمدند شباهت کلی به نقشههای فعالسازی استاندارد داشتند. با این حال نقشههای انسجام مناطق خیلی مهمی از فعالسازی را نشان دادند که با استفاده از روش استاندارد در روش 16/12 مورد دیده نمیشود. این نقشهها شامل مکان مناطق زبان در مجاورت تومورهای مغزی بودند که یافتههای روش انسجام با تحریک مستقیم قشری حین عمل جراحی تأیید شدند. بهبود نقشههای پاسخ وظیفه براساس تصویربرداری راکتیویته عروقی از لحاظ آناتومیکی فعالسازی دقیقتر و قویتر بویژه در مجاورت تومورها را در مقایسه با نقشههای به دست آمده از اطلاعات راکتیویته عروقی نشان داد.
نتیجهگیری: نتایج اولیه بدست آمده اصلی را نشان میدهد جداشدگی عصبی عروقی که اثر آن بر صحت BOLD fMRI در مجاورت تومورهای مغزی شناخته شده است، حداقل تا حدی امکان دارد با ترکیب یک اندازهگیری مستقل از راکتیویته عروقی در تجزیه و تحلیل BOLD برطرف شود.
مقدمه
برش جراحي عصبی مهمترين گزینهی درمانی براي تومورهاي بدخيم مغزي است زيرا طول مدت و كيفيت بقا با حداكثر برش تومور افزایش مییابد [1،2]. بنابراین هدف جراحی تومور مغزی حداکثر رساندن برش تومور و درعین حال پرهیز از قشرهای سخنوری مهم مجاور است که برش ناخواسته آن میتواند منجر به عواقب ناگوار عصبی شود. به منظور حفظ عملکرد عصبی حیاتی که در نزدیکی تومور قرار دارد، جراحان مغز و اعصاب باید قادر به شناسایی محل آناتومیکی قشرهای سخنوری قبل یا حین عمل جراحی باشند.
در گذشته قشرهای سخنوری مانند قشر حرکتی با تحریک مستقیم قشر شناسایی شدهاند. بعدتر MRI عملکردی وابسته به سطح اکسیژن خون (BOLD fMRI) بطور موفقیتآمیز در برنامهریزی و انجام برش تومورهای مغزی مورد استفاده قرار گرفته است [3،4]. با این حال، پی بردند که نقشههای BOLD مجاور تومورهای مغزی درستی و صحت محدودی داشتهاند [5-9]. fMRI BOLD بر این فرض استوار است که بین فعالیتهای نورونی و جریان خون یک اتصال وجود دارد. با این وجود رگهای جدید تومورهای بدخیم از لحاظ ساختاری و عملکردی[8] از جمله تغییرات در راکتیویته عروقی غیرنرمال هستند [14-10]. به دلیل اینکه BOLD fMRI پاسخ عروقی (به جای فعالسازی نورونی) را اندازه گیری میکند، در بیماران مبتلا به تومورهای بدخیم مغز پیدایش رگهای جدید غیر طبیعی تومور به افزایش فعالیت عصبی پاسخ نمیدهد که منجر به پاسخ خاموش BOLD میشود [15]. چند مطالعه اخیر روی موضع آناتومیکی جداشدگی عصبی عروقی کار کردهاند را که جداشدگی را با اندازهگیریهای کمی از راکتیویته عروق مغزی با نتایج fMRI BOLD منفی کاذب تعریف کردند [7، 16-18].
در نتیجه مدلهایی که یک تابع همودینامیکی یکنواخت به جای غیرنرمال را در سراسر مغز در نظر میگیرند، شاید برای تشخیص فعالسازی در یک قشر سخنوری تحت تأثیر تومور کافی نباشد. تجزیه و تحلیل دادهها براساس توابع یکنواخت پاسخ همودینامیکی احتمالاً به از دست رفتن فعالسازی (یا خطاهای منفی کاذب) در نقشههای پارامتری آماری BOLD منجر شود.
یکی از راهکارهای ممکن جهت برطرف کردن محدودیت نتایج منفی کاذب که از وجود عروق جدید غیرنرمال تومور و جدا شدگی عصبی عروقی حاصله بود میآید این است که راکتیویته عروقی را بصورت مستقل اندازه گیری کرده و این دادهها را در تجزیه و تحلیل BOLD fMRI ادغام کرد. یکی از شیوههای اندازهگیری مستقل راکتیویته عروقی حبس کردن تنفس است، که منجر به کمبود اکسیژن و افزایش کربن دیاکسید میشود و تحت شرایط نرمال منجر اتساع عروقی عروق مغزی میشود [14،17،18]. این تغییرات در اتساع عروقی را میتوان با استفاده از fMRI اندازهگیری کرد زیرا پاسخهای fMRI BOLD به پارادایمهای روتین وظیفه اندازهگیری میشوند. فرض این است که تابع پاسخ همودینامیکی عروق جدید غیرطبیعی صرف نظر از محرک (fMRI حبس کردن نفس یا وظیفه) از حالت نرمال تغییر میکند. اگر این فرض درست باشد، باید بتوان فعالسازی BOLD حتی در مناطق از پیدایش عروق جدید را با جستجو انسجام بین تابع پاسخ همودینامیکی در پارادایمهای fMRI حبس کردن تنفس و وظیفه روتین تشخیص داد.
هدف از این مطالعه استخراج یک مدل واکسلی است که همودینامیکهای تغییر یافته را با مقایسه نقشههای پاسخ BOLD بدست آمده حین انجام وظایف حرکتی و گفتاری با و بدون استفاده از وظیفه حبس کردن تنفس درنظر بگیرد. فرض میکنیم که نقشههای پاسخ BOLD به وظایف حرکتی و گفتاری که با این روش ترسیم میشوند به تصاویر فعالسازی استاندارد مدلهای یکنواخت پاسخ همودینامیکی شباهت کلی داشتند اما مناطق جدیدی از فعالسازی را نشان میدهند که بطور صحیحتری افزایش فعالیتهای عصبی را منعکس میکند و با روش استاندارد تجزیه و تحلیل fMRI BOLD به علت همودینامیکهای تغییر یافته تومورها مشخص نشده است.
مواد و روشکار
این پژوهش کاملاً مطابق با کد اخلاقی انجمن جهانی پزشکی (بیانیه هلسینکی) بود. بوردهای ناظر مؤسسهای مرکز سرطان Memorial Sloan-Kettering و Weill Cornell Medicine این مطالعه را تایید کردند. به دلیل ماهیت گذشتهنگر بودن این مطالعه نیازی به اخذ رضایت آگاهانه از بیمار نبود.
اطلاعات بیمار
هر بیمار تحت یک MRI آناتومی مغز و یک fMRI به عنوان بخشی از مراقبتهای روتین قبل از عمل جراحی قرار گرفتند. همه بیماران طی دو روز پس از MRI تحت عمل جراحی و برش تومور قرار گرفتند. آزمایشات پاتولوژیکی تومورها تشخیص گلیوبلاستوما چندشکلی (N = 9)، آستروسیتومای آناپلاستیک ( (N = 1، گلیومای درجه پایین (N = 5) یا متاستاز (N = 1) را نشان داد. برای مشاهده اطلاعات دموگرافیکی به جدول 1 مراجعه نمایید.
اسکن کردن fMRI و MRI عملکردی
در مجموع، 18 اسکن fMRI در 16 بیمار انجام شد. بیماران همچنین تحت MRI آناتومی مغزی در یک سیستم MRI بالینی 3.0 تسلا GEMS (Waukesha, WI) با کویل سر هشتی (8) شکل قرار گرفتند. حبس کردن نفس و fMRI وظیفهای با استفاده از fMRI echo-planar (TR = 4 s؛ TE = 40 ms؛ زاویه تکان 90 درجه؛ ماتریس 128 × 128 ؛ 240 mm FOV؛ ضخامت برش 4.5 میلی متر) انجام شد.
Abstract
Purpose
Preoperative functional MRI (fMRI) is limited by a muted BOLD response caused by abnormal vasoreactivity and resultant neurovascular uncoupling adjacent to malignant brain tumors. We propose to overcome this limitation and more accurately identify eloquent areas adjacent to brain tumors by independently assessing vasoreactivity using breath-holding and incorporating these data into the BOLD analysis.
Methods
Local vasoreactivity using a breath-holding paradigm with the same timing as the functional motor and language tasks was determined in 16 patients (9 glioblastomas, 1 anaplastic astrocytoma, 5 low grade astrocytomas, and 1 metastasis). We derived a model based on coherence for analyzing BOLD fMRI that takes into account the altered hemodynamics adjacent to brain tumors.
Results
Activation maps computed using the coherence model were overall similar to standard activation maps. However, the coherence maps demonstrated clinically meaningful areas of activation that were not seen using the standard method in 12/16 cases. This included localization of language areas adjacent to brain tumors, where the coherence method results were confirmed by intra-operative direct cortical stimulation. Enhanced task response maps based on vasoreactivity mapping demonstrated more robust, anatomicallycorrect activation, in particular adjacent to tumors as compared to maps obtained without vasoreactivity information.
Conclusions
The present preliminary results demonstrate the principle that the neurovascular uncoupling known to affect the accuracy of BOLD fMRI adjacent to brain tumors may be, at least partially, overcome by incorporating an independent measurement of vasoreactivity into the BOLD analysis.
INTRODUCTION
Neurosurgical resection remains the most important treatment option for malignant brain tumors as both the length and quality of survival are improved with maximized tumor resection [1,2]. Therefore, the goal of brain tumor surgery is to maximize the resection of the tumor while avoiding important adjacent eloquent cortices, whose inadvertent resection can lead to devastating neurological consequences. In order to preserve vital neurological function located near the tumor, it is important for the neurosurgeon to be able to identify the anatomical location of the eloquent cortices, either pre- or intraoperatively.
Traditionally, the eloquent cortices, such as the motor cortex, have been identified by direct cortical stimulation. More recently, blood-oxygen-level-dependent functional MRI (BOLD fMRI) has been used successfully in planning and carrying out the resection of brain tumors [3,4]. However, it has been found that BOLD maps adjacent to brain tumors have limited accuracy [5- 9]. BOLD fMRI is based on the premise that there is a coupling between neuronal activity and blood flow. However, the neovasculature of malignant tumors is known to be abnormal both structurally and functionally [8], including changes in vascular reactivity [10-14]. Since BOLD fMRI measures the vascular response (rather than neuronal activation), in patients with malignant brain tumors the abnormal tumor neovascularity does not respond as vigorously to increased neuronal activity, which leads to a muted BOLD response [15]. A number of recent studies have correlated the anatomical location of neurovascular uncoupling, defined by quantitative measurements of cerebrovascular reactivity, with false negative BOLD fMRI results [7,16-18].
As a consequence, models assuming a uniform rather than an abnormal hemodynamic response function over the whole brain may not be sufficient to detect activation in an eloquent cortex influenced by the tumor. Data analysis based on the uniform hemodynamic response functions may lead to missed activation (or false negative errors) in the BOLD statistical parametric maps.
A possible way to overcome the limitation of false negative results caused by the presence of abnormal tumor neovasculature and resultant neurovascular decoupling is to independently measure vasoreactivity and to incorporate these data into the BOLD fMRI analysis. One way to independently measure vasoreactivity is by breath holding, which leads to hypoxia and hypercapnia, which under normal circumstances leads to vasodilatation of the cerebral vasculature [14,17,18]. These changes in vasodilatation can be measured by fMRI in the same way as BOLD fMRI responses to routine task paradigms are measured. The assumption is that the hemodynamic response function of abnormal neovasculature is altered from the norm in the same way irrespective of the stimulus (breath-holding or task fMRI). If this assumption holds true, then one should be able to detect BOLD activation even in areas of abnormal neovasculature by searching for coherence between the hemodynamic response function in the breath-holding and routine task fMRI paradigms.
The purpose of this study is to derive a voxel-specific model that takes into account altered hemodynamics by comparing BOLD response maps obtained during motor and language tasks with and without the incorporation of a breath-holding task. We hypothesize that BOLD response maps to motor and language tasks computed this way will be overall similar to standard activation maps based on uniform hemodynamic response models but will also show new areas of activation that accurately reflect increase in neuronal activity that were not detected by the standard method of BOLD fMRI analysis due to altered hemodynamics of the tumors.
MATERIALS AND METHODS
The research performed was in full compliance with the Code of Ethics of the World Medical Association (Declaration of Helsinki). The institutional review boards of Memorial SloanKettering Cancer Center and Weill Cornell Medicine approved the study. Informed Consent was not obtained, as the study was a retrospective review.
Patient information
Each patient underwent an anatomical brain MRI and an fMRI as part of their routine preoperative care. All underwent resection of the tumor within two days of the MRI. Pathological examination of the resected tumors revealed the diagnosis of glioblastoma multiforme (N = 9), anaplastic astrocytoma (N = 1), low-grade glioma (N = 5), or metastasis (N = 1). See Table 1 for demographic data.
fMRI and anatomical MRI scanning
In total, 18 fMRI scans were performed on 16 patients. Patients also underwent an anatomical brain MRI on a 3.0 Tesla GEMS (Waukesha, WI) clinical MRI system with an 8-channel head coil. Breath-hold and task-specific fMRI were performed using echo-planar fMRI (TR = 4 s; TE = 40 ms; 90o flip angle; 128 × 128 matrix; 240 mm FOV; 4.5 mm slice thickness).
چکیده
مقدمه
مواد و روشکار
اطلاعات بیمار
اسکن کردن fMRI و MRI عملکردی
تحلیل دادههای fMRI
1. تجزیه و تحلیل استاندارد بر اساس ضرایب همبستگی
2. تحلیل استاندارد اصلاح شده براساس انسجام
3. تحلیلی انسجامی که ارزشیابی راکتیویته عروقی را از دادههای حبس کردن تنفس بدست میآورد
یافتهها
رابطه میان پاسخ عمل دستوری و پاسخ حبس کردن نفس
مقایسه روش استاندارد تجزیه و تحلیل و روش منسجم پیشنهادی
مثال 1
مثال 2
مثال 3
خلاصه
درجه حرکت در پویشهای حبس کردن نفس و وظیفه دستوری
بحث
نتیجهگیری
منابع
Abstract
INTRODUCTION
MATERIALS AND METHODS
Patient information
fMRI and anatomical MRI scanning
fMRI data analysis
1. Standard analysis based on correlation coefficients
2. Modified standard analysis based on coherence
3. Coherence based analysis incorporating vascular reactivity assessment from the breath-hold data
RESULTS
Relationship between the task response and the breath-hold response
Comparison of the standard method of analysis and the proposed coherence method
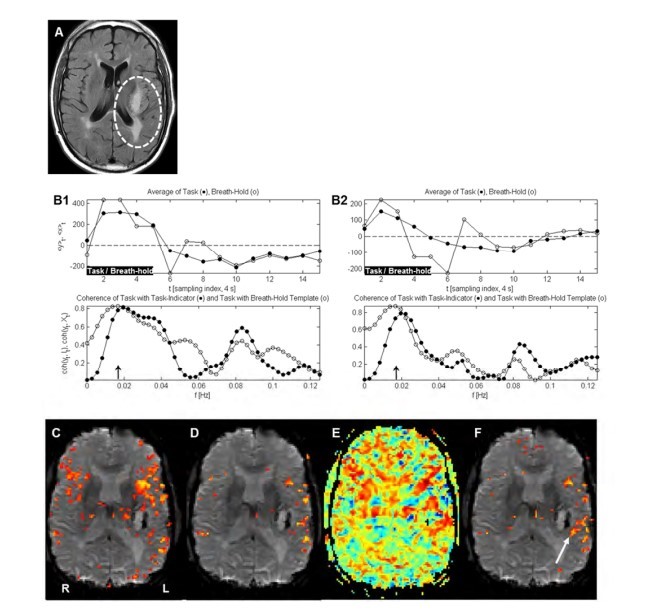
Example 1
Example 2
Example 3
Summary
Degree of motion in the breath-hold and the task scans
DISCUSSION
CONCLUSION
REFERENCES
