
دانلود رایگان مقاله دیدگاه پزشک در مورد مراقبت های اولیه در ارائه واکسن بزرگسالان
چکیده
به تازگی، چند واکسن جدید برای بزرگسالان توصیه می شود. کمی در مورد خرید واکسن سازی و شیوه های ذخیره مراقبت های اولیه پزشکان بزرگسالان شناخته شده است. برای تعیین نسبت عمل خانواده و پزشکان طب داخلی که به طور معمول واکسن خاص بزرگسالان را ذخیره می کند و منطق خود را برای کسانی که تصمیم به خرید واکسن دارند بیان می کند، ما یک مطالعه مقطعی در سال 2009 از یک نمونه تصادفی ملی از 993 پزشک خانواده (FPS) و 997 پزشک امراض داخلی عمومی (IMS ) در امریکا انجام دادیم. از 1109 پاسخ دهندگان 886 گزارش دادند که آنها مراقبت های اولیه برای افراد بزرگسال بالای 19-64 سال انجام می دهند و 96 درصد از این کالای پزشکان ارائه دادند که حداقل یک واکسن برای هر بزرگسال توصیه می شود. از این تعداد، 2٪ طرح متوقف واکسن و 12٪ طرح افزایش خرید واکسن؛ بقیه طرح حفظ وضع موجودی انبار را نظر دادند. از پاسخ دهنگان 27 ٪( EPS 31٪ در مقابل 20٪ Ms) همه واکسن بزرگسالان را ذخیره می کنند. نتیجه می گیریم که بسیاری از پزشکان که مراقبت های اولیه را برای بزرگسالان ارائه می دهند، همه واکسن توصیه شده را انبار نمی کنند. تلاش ها جهت بهبود میزان ایمنی سازی بزرگسالان انجام شود تا این موضوع اساسی را بررسی کنند.
1. مقدمه
اگر چه قوانین ایمن سازی به بخشی از مراقبت پیشگیرانه برای کودکان روتین تبدیل شده است، موارد مشابه برای بزرگسالان درست نیست. بر فرض مثال، تنها 17 درصد از غیر سالمند (کمتر از 65 سال ) بزرگسالان در معرض خطر، این واکسن تجویز شده پنوموکوک [1[ را دریافت کرده اند. مطالعات متعددی جهت بررسی عدم دریافت تعداد بیشتری از واکسن توسط بیماران بالغ انجام شده است. بیماران اغلب به پزشک خود گزارش کرده اند. به طور فعال واکسن توصیه نمی کنند و مفروضات اشتباه در مورد نیاز خود را برای ایمن سازی شناسایی کرده اند [2.[ ارائه دهندگان اغلب مسائل مختلف مطرح شده توسط بیماران، از جمله نگرانی های بیمار با توجه به عوارض جانبی، ترس بیمار از سوزن و عدم پوشش بیمه به عنوان دلایل میزان کم ایمنی سازی در شیوه های خود [2 [شناسایی کرده اند. مطالعات دیگر انواع عوامل مربوط به میزان پایین واکسیناسیون بزرگسالان مانند عدم ارائه مراقبت های اولیه به طور منظم [3 [به طور بالقوه سبک زندگی گیج کننده و یاعلائم مبتنی بر شرط برای برخی از واکسن ها [4 [ و عدم اولویت بندی از جوامع حرفه ای پزشکان که مراقبت برای بزرگسالان فراهم میکند.
روش معمول اطلاع رسانی پزشکان که مراقبت های پیشگیرانه برای بزرگسالان بی بهره از برنامه ایمن سازی نیز را ارائه می دهند. اگر چه یک برنامه ایمن سازی مرکب دوران کودکی سالانه در ژورنال پزشکی کودکان حداقل برای چند دهه گذشته منتشر شده است ، آن را تا اواخر سال 2007 نبود که سالانه پزشک داخلی اولین برنامه بزرگسالان ملی اعلام و در حال حاضر این کار سالانه در صفحات آن انجام می شود [[5 چنین تلاش هایی تا حد زیادی مورد نیاز است همانطورکه بسیاری از پزشکان به بیماران خود توصیه نمی کنند چرا که آنها از توصیه های ایمن سازی بزرگسالان فعلی آگاه نیستند. [2] در طول چند سال گذشته، چندین واکسن های جدید برای بزرگسالان توصیه شده است. ارائه این واکسن ها و تنظیمات آن به پزشک نیازخواهد داشت تا این واکسن ها را به شیوه های خود ذخیره کنند. در مقابل بسیاری از داروها یا بیولوژیک توسط پزشکان دیگر تجویز شده است، واکسن باید به طور مستقیم با شیوه های پیش از تقاضای بیمار خریداری شود. این نیاز به یک مبلغ سرمایه گذاری مالی در بخشی از این عمل برای خرید واکسن دارد که این عمل ممکن است، یا ممکن نیست، قادر به فروش باشد. هرچه تعداد از انواع محصولات واکسن و تعداد دوز خریداری می شد، هزینه پیش پرداخت زیادی بدون قطعیت از فروش مجدد مورد نیاز است، بیشتر هزینه های آتی مورد نیاز بی شک از فروش مجدد. حاصل خواهد شد حعلاوه بر این، واکسن نیاز به انبار خنک دارند و این در عوض نیاز به سرمایه گذاری در یخچالهای خاص و هشدار دما است.
اگرچه پزشکان متخصص اطفال به انبار کردن واکسن های متفاوتی عادت کرده بودند، تعداد کمی از پزشکان در مورد خرید واکسن و شیوه های انبارداری آن آگاه هستند که مراقبتهای اولیه برای بزرگسالان ارائه می دهند. چنین شیوه هایی بخش مهمی از ایجاد محیط در دسترس بودن واکسن برای بیماران بزرگسال است. ما جستجو کردیم تا سهم داروی داخلی و پزشک خانواده را تعین کنیم که بطور مداوم واکسن های خاص بزرگسالان و منطقشان را برای تصمیمات مذکور انبار کنند.
2. شیوه ها
2.1 نمونه
ما نمونه تصادفی ملی را از 1000 خانواده پزشک (FPS) و 1000 پزشک داخلی عمومی (OMs) از انجمن پزشکی آمریکا (AMA)، پزشکان از طریق فروشندگان پیمانکار استخراج کردیم. پزشکان ارشد انجمن پزشکی آمریکا دارای جامع ترین پایگاه داده پزشکی دارای مجوز طبابت در آمریکا هستند و شامل هردو عضو و غیر عضو انجمن پزشکی آمریکا است. نمونه گیری ما شامل کلیه پزشکان داروهای آلوپاتی (MD) و استخوان (DO) پزشکان خود-توصیفی به عنوان پزشک خانواده یا پزشک داخلی عمومی مبتنی بر مطب، برای مریضان تحت درمان مستقیم می شود. مستثنی پزشکانی با هرگواهی فوق تخصص صادره از هیئت مدیره سن بیشتر یا مساوی 70 سال، در حال حاضر در آموزش رزیدنسی یا استخدام شده داری تسهیلات پزشکی شخصی، بصورت فدرال (به عنوان مثال، Veterans Affairs به عنوان مثال، امور سربازان بازنشسته ) می شود. پس از بررسی 2000 پرونده در نمونه پزشکان ارشد انجمن پزشکی آمریکا، ما مستثنی کردیم . 7FPS و 3IMs که معیارهای ما در این آزمون را برآورده نکردند.
2.2 طرح نظرسنجی
ابزار گرداآوری اطلاعات ما پرسشنامه 4-صفحه، شامل 15 سوال می باشد، آن بررسی کرد که آیا پاسخ دهندگان در حال حاضر در انبار هر گونه واکسن برای بزرگسالان از سن 19 تا 64 سالگی طبابت می کنند و اگرنه، دلایلی برای انبارنکردن هر گونه واکسن برای این گروه است. سپس برای هر کدام از 10 واکسن مرتبط با بزرگسالان از سن 19 تا 64 سالگی، نظرسنجی انجام شد که آیا طبابت کردن با موجودی های انبار که واکسن برای بزرگسالان از سن 19 تا 64 سالکی و اگرنه، دلایلی برای انبارنکردن که واکسن برای این گروه وجود ندارد. 10 واکسن عبارتند از:
هپاتیت A؛ هپاتیت B؛ واکسن برای ویروس پاپیلومای انسانی (HPV)؛ دانه های سرخک، اوریون و سرخجه (MMR): واکسن کونژوگه مننگوکوکی (MCV4)؛ پلی ساکارید پنوموکوکی (PPSV23)؛ دیفتری کزاز (TCI)؛ ترکیب کزاز، دیفتری، سیاه سرفه و (TDAP)؛ آبله مرغان؛ و زوستر.
پرسش های دیگر که نگرش مخاطبان را در مورد بازپرداخت هزینه و تجویز واکسن برای افراد بالای 19-64 سال؛ بررسی کرد، طرح طبابت در سال آینده برای انبار کردن واکسن برای سالمندان 19-64 ساله (توقف، افزایش، کاهش / تعداد مشابه از واکسن های مختلف) خواهد بود که آیا طبابت با یک گروه خرید یا تعاونی خرید برای واکسن بزرگسالان شرکت؛ و ویژگی های شیوه یا طبابت شرکت می کند.
سوال اضافی در مورد دخالت تصمیم گیری پاسخ دهندگان در عمل یا طبابت با توجه به خرید واکسن، درخواست "تا چه حد شما در تصمیم گیری در مورد خرید واکسن برای طبابت خود درگیر هستید؟" پاسخ دهندگان گزارش دادند که آنها "به طور مستقیم در تصمیم گیری خرید واکسن درگیر هستند" به عنوان تصمیم گیرندگان (DMS) تعریف شدند. کسانی که مدعی بودند " بطور غیر مستقیم درگیر" یا " اصلا درگیر نیستند" به عنوان غیر تصمیم گیرندگانDMS) ) طبقه بندی شدند.
هیئت داوری بین المللی دانشگاه میشیگان دانشکده پزشکی این مطالعه تایید شده است.
2.3. اجرای نظرسنجی
نامه نظرسنجی اولیه در پایان ماه آوریل 2009 به 1990 پزشکان (993 FPS، 997 IMS (فرستاده شد و شامل یک تعهدنامه نامه شخصی، ابزار نظرسنجی، و انگیزه نقدی 5 $ می باشد. ارسال دو نامه اضافی به غیر از پاسخ دهندگان حدودا در فواصل 4 هفته رخ داده است.
Abstract
Recently, several new vaccines have been recommended for adults. Little is known regarding the immunization purchase and stocking practices of adult primary care physicians. To determine the proportion of family practice and internal medicine physicians who routinely stock specific adult vaccines and their rationale for those decisions, we conducted a cross-sectional survey in 2009 of a national random sample of 993 family physicians (FPs) and 997 general internists (IMs) in the US. Of the 1109 respondents, 886 reported that they provide primary care to adults aged 19–64 years and 96% of these physicians stock at least one vaccine recommended for adults. Of those, 2% plan to stop and 12% plan to increase vaccine purchases; the rest plan to maintain status quo. Of the respondents, 27% (31% FPs vs 20% IMs) stocked all adult vaccines. We conclude that many primary care physicians who provide care to adults do not stock all recommended immunizations. Efforts to improve adult immunization rates must address this fundamental issue.
1. Introduction
Although the provision of immunizations has become a routine part of preventive care for children, the same is not true for adults. For example, only 17% of non-elderly (<65 years) high-risk adults have received the recommended pneumococcal vaccine [1].
Several studies have attempted to examine why adult patients do not receive immunizations in greater numbers. Patients frequently have reported their physician does not actively recommend vaccines and have identified mistaken assumptions regarding their own need for immunization [2]. Providers often identify different issues than those raised by patients, including patient concerns regarding side effects, patient fear of needles and lack of insurance coverage as reasons for low immunization rates in their practices [2]. Others studies have found a variety of factors contributing to low adult immunization rates such as the lack of a regular primary care provider [3], potentially confusing lifestyle or conditionbased indications for some vaccines [4], and lack of prioritization from professional societies of physicians who provide care for adults [4].
Routine methods of informing physicians who provide preventive care for adults of the immunization schedule also have been lacking. Although a combined childhood immunization schedule has been published annually in the journal Pediatrics for at least the past decade, it was not until late 2007 that the Annals of Internal Medicine first promulgated the national adult schedule and will now do so annually in its pages [5]. Such efforts are greatly needed as many physicians do not make recommendations to their patients because they are not aware of current adult immunization recommendations [2].
Over the past several years, there have been several new vaccines recommended for adults. Provision of these vaccines in private practice settings would require physicians to stock these vaccines in their practices. In contrast to many other pharmaceuticals or biologics prescribed by physicians, vaccines must be purchased directly by practices in advance of patient demand. This requires a financial outlay on the part of the practice to purchase vaccines that the practice may, or may, not be able to resell. The greater the number of types of vaccine products and numbers of doses purchased, the greater the up-front expenditure required without a certainty of resale. Additionally, vaccines require sensitive cold-chain storage and this, in turn, requires an investment in special refrigerators and temperature alarms.
Although pediatricians have long been accustomed to stocking many different vaccines, little is known regarding the immunization purchase and stocking practices of physicians who provide primary care to adults. Such practices are an essential part of creating an environment of vaccine availability to adult patients. We sought to determine the proportion of internal medicine and family physicians who routinely stock specific adult vaccines and their rationale for those decisions.
2. Methods
2.1. Sample
We drew a national random sample of 1000 family physicians (FPs) and 1000 general internists (IMs) from the American Medical Association (AMA) Physician Masterfile through a contracted vendor. The AMA Physician Masterfile is the most comprehensive database of physicians licensed to practice in the United States, and includes both AMA members and non-members. Our sampling frame included all allopathic (MD) and osteopathic (DO) physicians self-described as a family physician or general internist in office-based, direct patient care. Excluded were physicians with any subspecialty board certification, age ≥70 years, currently in residency training, or employed at federally owned medical facilities (e.g., Veterans Affairs). After review of the 2000 records in the AMA Masterfile sample, we excluded 7 FPs and 3 IMs that were found to not meet our inclusion criteria.
2.2. Survey design
The 4-page, 15-item survey instrument addressed whether the respondent’s practice currently stocks any vaccines for adults aged 19–64 years and, if not, reasons for not stocking any vaccines for this group. Then for each of 10 vaccines relevant for adults aged 19–64 years, the survey asked whether the practice currently stocks that vaccine for adults aged 19–64 years and, if not, reasons for not stocking that vaccine for this group. The 10 vaccines were:
hepatitis A; hepatitis B; human papillomavirus vaccine (HPV); combined measles, mumps, and rubella (MMR); meningococcal conjugate vaccine (MCV4); pneumococcal polysaccharide (PPSV23); tetanus diphtheria (Td); combined tetanus, diphtheria, and pertussis (Tdap); varicella; and zoster.
Other questions addressed respondent attitudes regarding reimbursement for the cost and administration of vaccines for adults aged 19–64 years; the practice’s plans in the next year for stocking vaccines for adults aged 19–64 years (stop, increase/decrease/same number of different vaccines); whether the practice participates with a purchasing cooperative or buying group for adult vaccine; and practice characteristics.
An additional question targeted respondent decision-making involvement in the practice with respect to vaccine purchase, asking “To what extent are you involved in decisions about vaccine purchase for your practice?” Respondents who reported that they are “directly involved in vaccine purchase decisions” were defined as decision-makers (DMs). Those who said they were “indirectly involved” or “not involved at all” were classified as non-decisionmakers (nDMs).
The Institutional Review Board of the University of Michigan Medical School approved this study.
2.3. Survey administration
The initial survey mailing was sent at the end of April 2009 to 1990 physicians (993 FPs, 997 IMs) and included a personalized cover letter, the survey instrument, and a $5 cash incentive. Two additional mailings to non-respondents occurred at approximately 4-week intervals.
چکیده
1. مقدمه
2. شیوه ها
2.1 نمونه
2.2 طرح نظرسنجی
2.3. اجرای نظرسنجی
2.4 تجزیه و تحلیل داده ها
2.5. منبع مالی
3. نتایج
3.1 میزان پاسخ
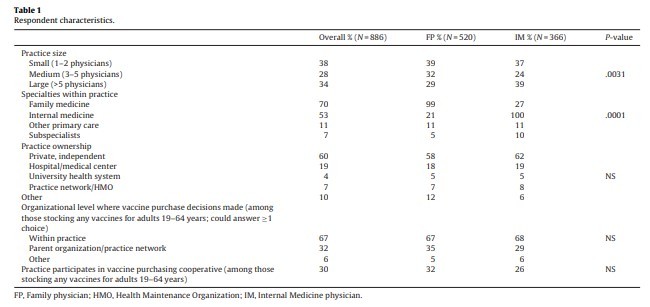
3.2 ویژگی های پاسخگو و طبابت
3.3. مسائل عمومی واکسن
3.4 ذخیره واکسن های خاص
4. بحث
منابع
Abstract
1. Introduction
2. Methods
2.1. Sample
2.2. Survey design
2.3. Survey administration
2.4. Data analysis
2.5. Funding source
3. Results
3.1. Response rate
3.2. Respondent and practice characteristics
3.3. General vaccine issues
3.4. Stocking of specific vaccines
4. Discussion
References
