
دانلود رایگان مقاله تکثیرپذیری تغییر سیگنال BOLD حاصل از نگه داشتن دم
چکیده
هم سنجی وابسته سطح اکسیژن خون (BOLD) تحت تاثیر برخی از عوامل فیزیولوژیکی است مانند جریان خون و حجم خونی که می تواند منبعی از تغییرات در آنالیز fMRI باشد. مطالعات قبلی پیشنهاد دادند که از داده های پاسخ مغزی برای کالیبراسیون و نرمالایز کردن نگاشت های BOLD در جهت کاهش تغییرات داده های fMRI در میان نواحی مغزی در آنالیز فردی و در سراسر افراد استفاده کنیم. نگه داشتن دم یکی از گسترده ترین روشهای مورد استفاده برای بررسی واکنش پذیری عروقی است. با این حال، قدرت و تکثیرپذیری این روش به خوبی شناخته نشده است. در این مطالعه به بررسی سه دوره نگه داشتن دم می پردازیم. از افراد خواسته شد که نفس خود را برای 9، 15 و 21 ثانیه در سه مرحله مجزا نگه دارند و موافقت نامه fMRI بعد از 15 تا 20 روز تکرار شد. داده های ما نشان می دهد که BOLD به نگه داشتن دم بعد از نتایج حاصل از یک شکل پیچیده پاسخ می-دهد که دلیل آن عوامل فیزیولوژیکی است که بر روی تغییر سیگنال با زمانی که بشدت تکثیرپذیر است تاثیر می-گذارند. با این وجود، تکثیرپذیری مقدار پاسخ مغزی به دی اکسید کربن بصورت دامنه سیگنال BOLD و تعداد واژگان پاسخ بیان می شود که به شدت به مدت زمان دوره های نگه داشتن دم وابسته است. دوره نگه داشتن دم 9 ثانیه ای منجر به تغییر شدید مقدار پاسخ می شود در حالیکه دوره های طولانیتر پاسخهای BOLD تکثیرپذیر و محکمی را تولید می کنند.
مقدمه
تصویرسازی تشدید مغناطیسی کارکردی (fMRI) بر اساس هم سنجی وابسته به سطح اکسیژن خون (BOLD) بطور وسیعی برای نشان دادن فعالیت مغز در افراد سالم و بیمار استفاده شده است (اگاوا، 1990). این روش از مدولاسیون جریان خون مغزی ناحیه ای در پاسخ به فعالیت عصبی بهره می برد (جیروارد و لادکولا، 2006). با این حال، این یک فرآیند پیچیده است و ارتباط بین پاسخ BOLD و متابولیسم عصبی بطور جزیی شناخته شده است زیرا پاسخهای BOLD تحت تاثیر هر تغییر همودینامیک مغزی قرار دارند (لوگوتتیس، 2002). برای مثال، گزارش شده است که تغییرات حالت عروقی پایه به دلیل مواد واسواکتیو (مانند کافیین) (مولدرینک، 2002)، عوامل دارویی (برون، 2003) سن و بیماریهای مغزی (وارد، 2008) می تواند پاسخ BOLD را دچار تغییر کند. حتی تغییر خودبخودی الگوی نفس کشیدن (وایز، 2004) یا دوره های کوتاه نگه داشتن دم (BH) نیز منجر به تغییرات CBF می شود و ممکن است منبع تغییر سیگنالهای مدلسازی نشده در مطالعات fMRI باشد (ابوت، 2005). علاوه بر این، اشاره شده است که پاسخ سیگنال BOLD به این عوامل در مناطق مغزی همگن نیست (ایتو، 2000)، که منجر به تغییری می شود که قدرت آماری را در آنالیز fMRI کاهش می دهد و باعث پیچیده شدن تفسیر نتایج فرد می شود (هندورکر، 2004). پیشنهاد شده است که حداقل بخشی از این تغییر می تواند زمانی که اطلاعات واکنش پذیری مغزی در تحلیل با هدف شناسایی پاسخهای BOLD به دلیل جفت شدن عصبی-روانی مدلسازی می شود به حساب آید. تصدیق این فرض، باندتینی و وانگ (1997) نشان دادند که نگاشته های fMRI BOLD می توانند در میان نواحی مغزی با استفاده از واکنش پذیری عصبی به استنشاق دی اکسید کربن نرمالایز شوند. یک روش مشابه نیز توسط کوهن (2004) ارائه شده است که قادر به کاهش قابل توجه اختلافات در نگاشته های fMRI BOLD حاصل در قدرتهای مختلف میدان تشدید مغناطیس بود. دیویس و همکاران (1998) این روش را با ارائه روشی بر اساس واکنش پذیری مغزی-عروقی ناشی از استنشاق دی اکسید کربن بعنوان مرجعی برای کالیبراسیون BOLD fMRI به منظور استخراج نرخ متابولیکی کمی مغزی مقادیر اکسیژن (CMRO2) توسعه دادند.
نگه داشتن دم بعنوان یک روش جایگزین برای استنشاق دی اکسید کربن در جهت ایجاد نگاشته های واکنش-پذیری مغزی ارائه شده است (کورفیلد، 2001؛ کاستراپ، 1999؛ لی، 2000). تحت این فرض که CMRO2 بدون تغییر باقی می ماند، BH منجر به کاهش اکسیژن و تجمع دی اکسید کربن در جریان خون می شود. افزایش دی اکسید کربن در خون باعث اتساع عروق مغزی و افزایش CBF می شود (مارکوس و هریسون، 1992). کاستراپ و همکاران (2001) شباهت بین کار BH و استنشاق دی اکسید کربن در ارزیابی ظرفیت ذخیره سازی همودینامیک با BOLD fMRI را نشان داد. با این حال، روش BH ساده تر است و ضرر کمتری نسبت به استنشاق دی اکسید کربن دارد. بنابراین، می تواند در هر زمینه آزمایشگاهی یا درمانی به کار برده شود. مطالعات قبلی نشان دادند که پاسخهای مهم BOLD می توانند در طول BH با مدت زمان کوتاه 10 ثانیه اندازه گیری شوند (لیو، 2002). کاستراپ و همکاران (1998) زمانهای مختلفی را برای نگه داشتن دم (18، 30 و 40 ثانیه) مورد مطالعه قرار دادند و تغییرات سیگنال BOLD را بین 8/0 و 5/3 درصد گزارش کردند. توماسون و همکاران (2005) قادر به استفاده کردن از نگه داشتن دم حتی در کودکان بودند، که نشان دهنده اختلافات عمده در نگاشته های واکنش پذیری مغزی بین کودکان و بزرگسالان بود. از این رو، بطور کلی شواهد زیادی بر روی اثربخشی نگه داشتن دم برای ایجاد نگاشته های واکنش پذیری مغزی عصبی وجود دارد. در واقع، توماسون و همکاران (2007) بطور موفقیت آمیزی روش کالیبراسیون را با استفاده از نگه داشتن دم برای روبش واکنش پذیری مغزی فردی به کار بردند.
به منظور استفاده از نگاشته های واکنش پذیری مغزی BOLD در جهت کالیبراسیون یا نرمالیزاسیون داده های fMRI این ضرورت وجود دارد که بدانیم این نگاشته ها به چه مقدار قابل اطمینان هستند. یک راه برای ارزیابی قابلیت اطمینان روش عبارتست از تخمین تکثیرپذیری آن تحت برخی از شرایط. برای اطلاع ما، تاکنون هیچ مطالعه ای به بررسی تکثیرپذیری پاسخ BOLD ناشی از BH نپرداخته است. برخی از مطالعات تکثیرپذیری تغییرات سیگنال BOLD ناشی از کارهای حرکتی را (هاول، 2006؛ لیو، 2004)، کارهای حسی (پیلن و دوینگ، 2005) و پارادایم شناختی (چی، 2003؛ فرناندز، 2003؛ رائو، 2007) بررسی کردند.
بیشترین روش استفاده شده برای تخمین تکثیرپذیری پاسخ BOLD عبارتست از همبستگی بین اندازه گیریهای تکرار شده بصورت ضریب همبستگی رایج محصول پیرسون (فرناندز، 2003) یا آنالیز همبستگی درون رده ای (ICC) (وی، 2004؛ راماکرز، 2007). ضریب همبستگی پیرسون قدرت یک رابطه بین دو متغیر را اندازه گیری می کند اما نسبت به مقادیر مطلق تغییر هیچ حساسیتی ندارد. بنابراین، اطلاعاتی را در مورد توافق بین دو اندازه-گیری در اختیار قرار نمی دهد. ICC استفاده شده در مطالعات fMRI برای بیان اینکه چقدر فعالسازی هر فرد نسبت به گروه دیگر در طول مراحل مختلف سازگار است استفاده می شود (زاندبلت، 2008). انواع مختلف روشهای ICC باید بسته به چگونگی تخمین واریانس اندازه گیری ها به کار برده شوند (برای مثال، آنالیز و تحلیل واریانس یک طرفه در مقابل تحلیل دوطرفه). هر روش ICC ممکن است نتایج مختلفی را ایجاد کند و روشهای مختلف برای ارزیابی تکثیرپذیری در fMRI پیاده سازی شده است (فرناندز، 2003؛ اسپچ، 2003). علاوه بر این، هر دو روش ICC و پیرسون نسبت به گسترش مقادیر در نمونه حساس هستند (بلاند و آلتمن، 1990). از آنجا که روشهای همبستگی اطلاعات بیشتری را در مورد روند تغییر نسبت به تکرارپذیری اندازه گیری ها در اختیار قرار می دهند، روشهای اضافی باید برای تخمین تکثیرپذیری اندازه گیری در نظر گرفته شوند. ضریب اختلاف (CV) بیان شده بصورت نسبت بین انحراف استاندارد و میانگین، بعنوان شاخص تکثیرپذیری اندازه گیری پیشنهاد شده است (مارشال، 2004؛ جاندرا، 2005) و در واقع برای ارزیابی تکثیرپذیری نگاشته های واکنش پذیری مغزی ناشی از استنشاق دی اکسید کربن مورد استفاده قرار گرفته است. مزیت عمده آن این است که یک اندازه گیری ثابت مقیاسی از نابرابری را ارائه می دهد.
برای ارزیابی تکثیرپذیری سیگنال BOLD، پارامترهای مختلفی باید در نظر گرفته شوند. تا به امروز، هر مطالعه ای در مورد روی تکثیرپذیری تنها بر روی برخی از جوانب پاسخ BOLD مانند پویایی زمانی پاسخ (نیومن، 2003)، شدت سیگنال (رومبوت، 1998) اندازه فعالسازی و مدار t وکسل به وکسل تمرکز کرده اند (فرناندز، 2003). در این مقاله، ما به بررسی تنوع و تکثیرپذیری بسیاری از پارامترهای مختلف مرتبط با پویایی زمانی و مقدار پاسخ BOLD در میان افراد با استفاده از روش همبستگی پیرسون و ضریب اختلاف می پردازیم. ما به منظور شناسایی مناسبترین پارادایم BH برای حصول نگاشته واکنش پذیری مغزی BOLD قابل تجدید مدت زمان BH را دستکاری کردیم.
روشها
افراد
داده ها از میان افراد سالمی که رضایت آگاهانه خود را اعلام کرده بودند جمع آوری شد. چهار فرد از تحلیل حذف شدند. یک فرد به دلیل حرکت بیش از حد سر، یک فرد به دلیل نفس کشیدن خودبخودی در طول مرحله اسکن MR (خودگزارش وضعیت اضطراب) و دو فرد نیز به دلیل اجرا نکردن مرحله دوم اسکن حذف شدند. گروه آزمایشی متشکل از 11 فرد (5 مرد و 6 زن، سن: 3/8±7/31؛ محدوده 20 تا 42 سال) بود.
گرفتن MRI
تصاویر بر روی یک اسکنر 3 T MR (سیمنز، ارلانگن، آلمان) گرفته شدند. در طول هر عمل، 126 حجم مغزی با یک توالی EPI اکو (GE) اختلاف وزنی T2* (TR=3000 ms, TE=30 ms، زاویه تلنگر=90 درجه، ماتریس 64*64، FOV=192*192، تعداد برشها=36 قطعه قطعه، ضخامت قطعه=3میلی متر، فاصله شکاف= 10درصد، اندازه واکسل=3 میلی متر مکعب، پهنای باند خواندن در هر پیکسل 1980 کیلوهرتز) حاصل شد. تصاویر آناتومیک با وضوح بالا با استفاد از یک حجم مغزی سه بعدی وزنی T1 (TR=2300 ms, TE=3.93 ms، زاویه ضربه یا تلنگر=90درجه، رزولوشن 256*256 در صفحه، FOV=256، ضخامت برش=1 میلی متر، تعداد قطعات=160 قطعه) حاصل شد.
abstract
Blood oxygen level dependent (BOLD) contrast is influenced by some physiological factors such as blood flow and blood volume that can be a source of variability in fMRI analysis. Previous studies proposed to use the cerebrovascular response data to normalize or calibrate BOLD maps in order to reduce variability of fMRI data both among brain areas in single subject analysis and across subjects. Breath holding is one of the most widely used methods to investigate the vascular reactivity. However, little is known about the robustness and reproducibility of this procedure. In this study we investigated three different breath holding periods. Subjects were asked to hold their breath for 9, 15 or 21 s in three separate runs and the fMRI protocol was repeated after 15 to 20 days. Our data show that the BOLD response to breath holding after inspiration results in a complex shape due to physiological factors that influence the signal variation with a timing that is highly reproducible. Nevertheless, the reproducibility of the magnitude of the cerebrovascular response to CO2, expressed as amplitude of BOLD signal and number of responding voxels, strongly depends on duration of breath holding periods. Breath holding period of 9 s results in high variability of the magnitude of the response while longer breath holding durations produce more robust and reproducible BOLD responses.
Introduction
Functional Magnetic Resonance imaging (fMRI) based on Blood Oxygen-Level Dependent (BOLD) contrast (Ogawa et al., 1990) has been extensively used to map brain activity both in healthy subjects and patients. The method exploits the modulation of the local cerebral blood flow (CBF) in response to neural activity (Girouard and Iadecola, 2006; Iadecola, 2004). However, this is a complex process and the relationship between the BOLD response and the neural metabolism is only partially understood since BOLD responses are affected by any cerebrovascular hemodynamic changes (Logothetis, 2002; Logothetis et al., 2001). For example, it has been reported that variations of the baseline vascular state due to vasoactive substances (e.g.: caffeine) (Mulderink et al., 2002), pharmacological agents (Brown et al., 2003), age or brain diseases (Ward et al., 2008; Wu and Hallett, 2005) can alter the BOLD response. Even spontaneous variation of breathing pattern (Wise et al., 2004) or brief periods of breath hold (BH) lead to CBF variations and may be a source of confounds or unmodelled signal variability in fMRI studies (Abbott et al., 2005). Furthermore, it has been noted that the BOLD signal response to these factors is not homogeneous among brain areas (Ito et al., 2000; Wise et al., 2004),
resulting in a variability that reduces the statistical power in group fMRI analysis and complicate the interpretation of single subject results (Handwerker et al., 2004). It has been suggested that at least part of this variability can be taken into account when the cerebrovascular reactivity information is modelled within the analysis aimed at identifying the BOLD responses due to neurovascular coupling. Verifying this assumption, Bandettini and Wong (1997) demonstrated that fMRI BOLD maps can be normalized among brain regions using cerebrovascular reactivity to CO2 inhalation. A similar approach has been proposed also by Cohen et al. (2004) who were able to significantly reduce differences in fMRI BOLD maps obtained at different MR field strengths. Davis et al. (1998) have extended this approach proposing a method based on cerebrovascular reactivity elicited by CO2 inhalation as a reference to calibrate BOLD fMRI in order to extract quantitative Cerebral Metabolic Rate of Oxygen (CMRO2) values.
Breath holding (BH) has been proposed as an alternative method to CO2 inhalation to generate cerebrovascular reactivity maps (Corfield et al., 2001; Kastrup et al., 1999a,b; Li et al., 1999, 2000; Liu et al., 2002). Under the assumption that CMRO2 remains invariant, the BHtask leads to a reduction of oxygen and an accumulation of carbon dioxide in the blood stream. The consequent hypercapnia induces cerebral vasodilatation and increased CBF (Markus and Harrison, 1992). Kastrup et al. (2001) showed similarity between BH task and CO2 inhalation in evaluating the hemodynamic reserve capacity with BOLD fMRI. However, the BH method is simpler and less invasive than CO2 inhalation. Thus, it could be applied in almost any clinical or experimental setting. Previous studies showed that significant BOLD responses could be measured during BH with duration as short as 10 s (Liu et al., 2002). Kastrup et al. (1998) studied different breath holding duration (18 s, 30 s, and 40 s) and report BOLD signal changes between 0.8% and 3.5%. Thomason et al. (2005) were able to use a breath hold task even in children, demonstrating significant differences in BOLD cerebrovascular reactivity maps between children and adults. Hence, overall there are many evidences on the efficacy of the breath hold task to generate cerebrovascular reactivity maps. In fact, Thomason et al. (2007) successfully applied the calibration procedure using breath hold to probe individual cerebrovascular reactivity.
In order to use BOLD cerebrovascular reactivity maps to calibrate and/or normalize fMRI data, however, it is necessary to know how much these maps are reliable. One way to assess the reliability of a method is to estimate its reproducibility under the same conditions. To our knowledge, so far no studies have explored the reproducibility of the BOLD BH-induced response. Some studies investigated the reproducibility of BOLD signal changes induced by motor tasks (Havel et al., 2006; Liu et al. 2004), sensory tasks (Peelen and Downing, 2005; Vlieger et al., 2003; Yetkin et al., 1996) and cognitive paradigms (Chee et al., 2003; Fernandez et al., 2003; Harrington et al., 2006; Rau et al., 2007).
The most used way to estimate the reproducibility of BOLD response is the correlation between repeated measurements computed as the Pearson product moment correlation coefficient (Fernandez et al., 2003; Jansen et al., 2006) or the intraclass correlation analysis (ICC) (Wei et al., 2004; Raemaekers et al., 2007). The Pearson correlation coefficient measures the strength of a relation between two variables but it is insensitive to the absolute values of the variation. Thus, it does not provide information about the agreement between two measures (Bland and Altman, 1986). The ICC applied to fMRI studies is used to express how consistent is the activation of each subject relative to the rest of the group over different sessions (Zandbelt et al., 2008). Different types of ICC methods must be applied depending on how the variance of the measurements is estimated (e.g., one-way vs. two-way analysis of variance) (McGraw and Wong, 1996). Each ICC method may produce different results, and different methods have been implemented to investigate the reproducibility in fMRI (Fernandez et al., 2003; Raemaekers et al., 2007; Specht et al., 2003). Furthermore, both ICC and Pearson correlation methods are sensitive to the spread of values in the sample (Bland and Altman, 1990). Since correlation methods give more information about the trend of variation than about the repeatability of the measures, additional methods must be considered to estimate the reproducibility of a measure. The coefficient of variation (CV), expressed as the ratio between the standard deviation and the mean, has been proposed as an index of reproducibility of a measure (Marshall et al., 2004; Tjandra et al., 2005) and in fact it has been used to evaluate the reproducibility of cerebrovascular reactivity maps elicited by CO2 inhalation;(Leontiev and Buxton, 2007). Its major advantage is that it represents a scale invariant measure of inequality (Allison, 1978).
Methods
Subjects
Data were collected from 15 healthy subjects who gave their informed consent. Four subjects were excluded from the analysis. One subject was excluded because of excessive head movement, one subject was excluded because his spontaneous breathing during the MR scanning session was irregular (self-report of state of anxiety) and two subjects did not perform the second scanning session. The experimental group was made up of 11 subjects (5 males, 6 females, age: 31.7 ±8.3; range 20–42).
MRI acquisition
Images were acquired on a 3.0 T MR head scanner (Siemens, Erlangen, Germany). During each functional run 126 brain volumes were acquired with a T2⁎-weighted Gradient Echo (GE) EPI sequence (TR = 3000 ms, TE = 30 ms, flip angle = 90°, 64 × 64 acquisition matrix, FOV = 192 ×192, number of slices =36 interleaved transversal, slice thickness = 3 mm, gap = 10%, voxel size = 3 mm3 , readout bandwidth per pixel 1980 kHz). High resolution anatomical images were obtained using a T1-weighted 3D brain volume (TR = 2300 ms, TE = 3.93 ms, flip angle = 90°, 256 × 256 in-plane resolution, FOV = 256, slice thickness = 1 mm, number of slices = 160 sagittal slices).
To investigate the reproducibility of the BOLD signal, different parameters may be taken into account. To date, each study on the reproducibility has focussed only on some aspects of the BOLD response such as the temporal dynamic of the response (Neumann et al., 2003), the signal intensity (Rombouts et al., 1998), the size of activation (Rombouts et al., 1998) and the voxel-by-voxel t-value (Fernandez et al., 2003). In the present work we investigate the variability and reproducibility of many different parameters related both to the temporal dynamic and to the magnitude of BOLD response within and across subjects using both the Pearson correlation method and the coefficient of variation. We manipulated the BH duration, in order to identify the most suitable BH paradigm to obtain a reproducible BOLD cerebrovascular reactivity map.
مقدمه
روشها
افراد
گرفتن MRI
توافقنامه fMRI
پیش¬پردازش اطلاعات FMRI
تحلیل داده¬ها
نتایج
آنالیز تغییرات سیگنال نگه داشتن دم
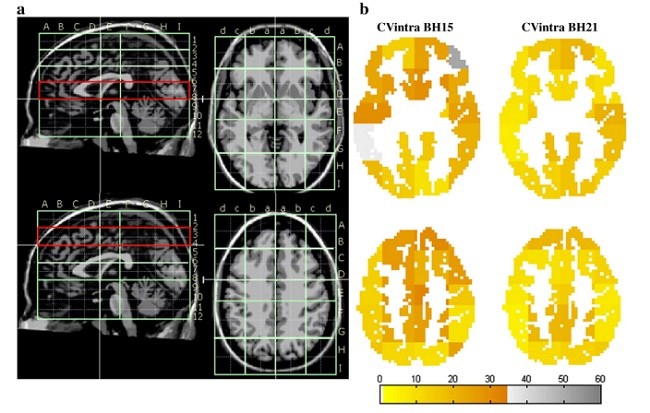
تکثیرپذیری نگه داشتن دم
بحث
Abstract
Introduction
Methods
Subjects
MRI acquisition
fMRI protocol
fMRI data pre-processing
Data analysis
Results
Analysis of breath holding signal changes
Breath holding reproducibility
Discussion
Acknowledgments
References
